Triassic lungs: Unidirectional flow in alligators’ breathing
The lungs of birds move air in only one direction during both inspiration and expiration through a set of tubular gas-exchanging bronchi called parabronchi. On the other hand, in the lungs of mammals and other vertebrates, air moves tidally into and out of terminal gas-exchange structures, called alveoli (Figure 1).

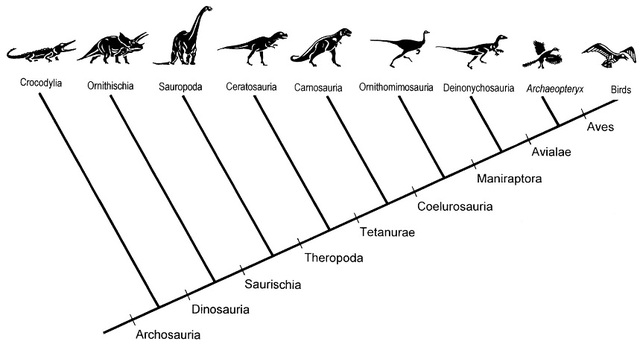
The work carried out by Farmer and Sanders 1 shows that unidirectional airflow is also present in the lungs of the American alligator. This fact suggests that such respiratory mechanism dates back to the Triassic Archosaurs, and not to the later coelurosaurs (Figure 2), hence being present in both dinosaur and nondinosaur descendants (phytosaurs, aetosaurs, rauisuchians, crocodylomorphs, and pterosaurs).
Bird respiratory system
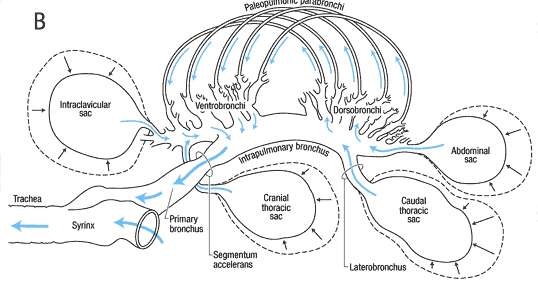
The avian respiratory system is different from that of other vertebrates, since it consists of small rigid lungs plus nine flexible air sacs. The air sacs act like bellows, not taking part in the actual gas exchange, but permitting a unidirectional flow of air through the lungs.
During inspiration (Figure 3, A), air from the trachea moves into the posterior air sacs and lungs, and through the parabronchi. Simultaneously, air from the lungs flows into the anterior air sacs.
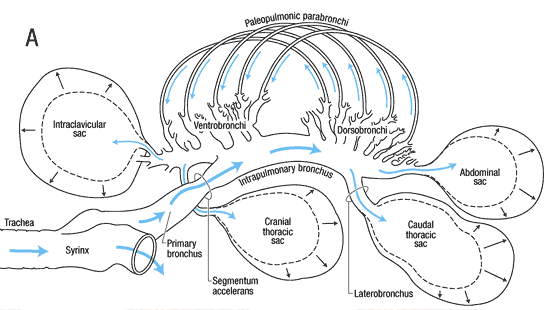
During exhalation (Figure 3, B), air from the posterior sacs moves into and through the lungs and parabronchi, and air from the anterior sacs flows to the trachea and out of the body. So, air always moves unidirectionally and, as a result, is higher in oxygen content than air in the alveoli of humans and other mammals, where new air is mixed with old air.
Alligator respiratory system
Crocodilian lungs are distinct from bird lungs and are thought to have an alveolar-arterial blood gas exchange. However, the topography of the intrapulmonary bronchus and of the first bronchi is similar in birds and crocodilians.
- The cervical ventral bronchus (CVB), pictured in green in Figure 4, is similar to the birds’ abdominal bronchus.
- The three dorsobronchi (blue in Figure 4) spiral toward the apex of the lung like in birds. Farmer and Sanders have also discovered that the dorsal bronchi connect to each other and to the CVB through numerous parabronchi with diameters between 1 and 1.5 mm.
- The small orifice to caudoventral bronchi (arrows, Fig. 1C) is placed opposite the orifice of the dorsal bronchi.
These anatomical similarities lead to the hypothesis that airflow in alligators might also be unidirectional. However, previous studies did not identify unidirectional flow in alligators’ lungs and this kind of flow was linked to the existence of abdominal air sacs and incompatible with the hepatic piston mechanism of ventilation of crocodilians.
Experiments
Three different sets of experiments were carried out to prove that airflow in alligator lungs is unidirectional. First, two flow-meters were implanted in the CVB (green in Figure 4) and in one of the dorsal bronchus (blue in Figure 4) of four alligators. Their lungs were then artificially ventilated, observing that air in the CVB moved in a cranial-to-caudal (head-to-tail) direction, while air in the dorsal bronchus moved in a ventrolateral (ventral part away from the midline) to dorsomedial (toward the back and near the midline) direction during both expiration and inspiration (Figure 4, F and G). Next, the airflow in the CVB during normal breathing was monitored in six alligators. And finally, the flow was also visualized by filling an excised lung with a saline solution that contained fluorescent microspheres. This last test also allowed the characterization of the flow in the parabronchi, confirming that it is unidirectional.

In short, all three methods confirm that airflow through the lungs of alligators is unidirectional as in birds, since the flow during the transition from inspiration to expiration continued rather than dropping to zero, as would be the case of a tidal flow.
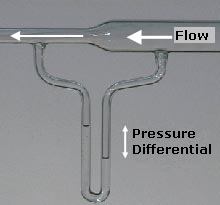
However, the experiments do not show how it is possible to have such flow in alligators, which don’t have air sacs and present diaphragmatic breathing. Figure 4 H and I show a mechanism for unidirectional flow that does not depend on air sacs or the mechanics of breathing. During exhalation, the geometry of the configuration makes the air flow through the upper part of the bifurcation; that is through the dorsobronchus, parabronchus and ventrobronchus. During inhalation the air is drawn into the lower part of the bifurcation. According to the authors, this is due to a decrease in pressure in this region caused by the Venturi effect (Figure 5). In my opinion it is merely a matter of mass conservation. However, I understand that the mechanism showed in Figure 4 H and I is a schematic of the real configuration; in which the effect mentioned by the authors may be dominant. This mechanism was tested in a glass model, proving that it gives rise to unidirectional airflow.
Conclusions
Previous works on the evolution of unidirectional airflow stated that it was first present in dinosaurs of coelurosaurian clade (Figure 2). Findings from the work by Farmer and Sanders show that the air sacs, which birds have and crocodilians lack, are not necessary to have unidirectional flow in the lungs. This suggests that such unidirectional flow was present in the basal archosaurs and their descendants. A more efficient mechanism of gas exchange like the one described in this work would have given archosaurs an evolutionary advantage during a period of relative lack of oxygen in the environment.
References
- C. G. Sanders, K. Farmer, Unidirectional Airflow in the Lungs of Alligators, Science 327, 338 (2010) ↩