Gene therapy safe and potentially effective against colour blindness
Gene therapy safe and potentially effective against colour blindness

Gene therapy was a 90s hit. The most promising medical advance of the decade was almost forgotten after some really serious side effects occurred in one clinical trial, but after nearly 3 decades a small trial 1 in Germany has shown gene therapy not only to be safe for use in humans but also to have benefits for people suffering from achromatopsia, a type of colour blindness.
Although we are more acquainted with partial colour blindness, like that affecting red or green, achromatopsia is a full colour blindness, and patients can only see in monochrome. This problem is caused in at least one third of cases by a mutation in a single gene, namely the gene CNGA3. And it is against this gene that the gene therapy from the collaboration between the LMU in Munich and the University Hospital in Tübingen was targeted.
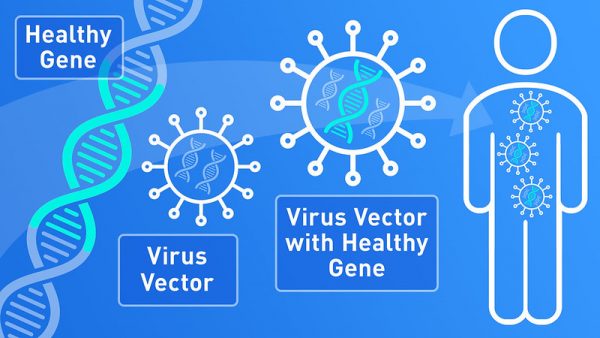
To get the healthy gene into the cells, a modified viral vector was used that although able to carry the gene inside the eye cells (cones), was unable of causing an infection. For this small trial, the gene therapy was injected into the most affected eye of 9 people (8 male, 1 female) and after a few weeks, they were re-evaluated for safety.
The researchers not only found the therapy to be completely safe, but also reported on an improvement of vision in terms of contrast, focus and most importantly in this case: colour vision.
However, this is still an early trial and more extensive ones with more enrolled patients and further endpoints need to be performed to consider the therapy a real option for patients. Moreover, this type of therapy should be best applied in childhood since then the brain has a higher degree of plasticity and can better create the new neural connections to accommodate for the new information retrieval, namely: colour.
That is why the researchers in this trial plan to include kids in the next phase of the trial. As good and promising as this all sounds especially for people with eye genetic diseases, there is yet another issue with the treatment. Even though in principle it would be enough with one application for a lifetime, gene therapy is expensive. So expensive that, for instance, Luxturna, an FDA-approved gene therapy for a rare genetic disease that can lead to blindness costs about 425,000 USD per eye!!!, and that is not even the most expensive gene therapy in the market.
References
- M.D. Fischer et al. (2020) Safety and Vision Outcomes of Subretinal Gene Therapy Targeting Cone Photoreceptors in Achromatopsia JAMA Ophtalmology doi: ↩
2 comments
[…] Mundua gris eskalan ikusten duenik bada, koloreak ikusteko gaitasuna ez duelako. Pertsona hauek ez dira daltonikoak, akromatopsikoak, baizik. Terapia genikoa izan daiteke hauen irtenbidea. Rosa García-Verdugoren Gene therapy safe and potentially effective against colour blindness […]
This is incredibly exciting news in the realm of gene therapy and its potential impact on vision disorders! I remember when my cousin, who has achromatopsia, told me about the difficulties he faces every day because he can’t perceive colors. The thought that a single treatment could change his life so fundamentally is both uplifting and awe-inspiring.