The perfume of chemokines: how cancer manipulates our defenses
The relation of cancer and the immune system has been traditionally regarded in terms of war; a force of courageous immune system soldiers representing, lets say, The United States of Your Body facing the evil forces of anarchy in the form of tumoral cells. Following this logic the immune system eradicates tumors (elimination phase) until tumors acquire enough growth rates to compensate the killings (equilibrium phase) and eventually outcompete the immune system (escape phase) 1. However, for the last twenty years there has been a change of paradigm. Now we know that for much killing the immune system inflicts in tumors, tumors use the immune system to grow! A terrifying paradox; the enemy uses our soldiers in its own benefit 2. And how? Well, this is the bad thing of having an enemy from your own body…They know all the dirty tricks.
Let´s go down to details; let´s talk about chemokines. Chemokines are small proteins that attract (among other cell types) immune cells expressing specific receptors against them. I like to think of chemokines as delicate perfumes attracting the rude soldiers of the immune system. But if this is so…Why so many cancer cells and cancer-associated cells secrete chemokines? What is the sense of using your worst enemy favorite perfume? The answer is that tumors attract immune cells with “Eau de Chemokines” and latter “seduce” them to start secreting growth factors, much appreciated by cancer cells. This double personality of some immune cells, that can kill or heal, is a natural part of the process of wound healing and cancer cells sequester this function to their own purpose. In fact, tumors have been compared to “wounds that do not heal”. In this way tumors influence the immune response against them making it chronic and benefiting from a continuous influx of immune cells that are latter corrupted and used for the tumor to grow.
Recently, the group of Joan Massague has provided an elegant and vastly accomplished work exemplifying the perverse abuse of the immune system by tumoral cells 3. First the authors explain the rationale for investigating the role of two chemokines, CXCL1 and CXCL2, in breast cancer. Around 7,5% of human breast tumors and, more interestingly, 20% of breast cancer metastasis, carry a gene amplification of these two chemokines (Figure 1). These results suggest that overexpression of CXCL1 and 2 contributes to breast cancer invasion and metastasis. To prove this concept the authors use different lines of breast cancer in which they silence the expression of CXCL1 and 2 with short hairpin RNAs. Afterwards they transplant control and CXCL1 or CXCL2 knock down cell lines in mice to obtain breast tumors. The results show that breast tumors grow less in lines with silenced CXCL1 and 2 compared to controls, and more interestingly, there are less and smaller lung metastases (Figure 1).

So far it is clear that these two chemokines are used by breast cancer cells to grow and metastasize; next question… How? First the authors analyze the tumors obtained before and they observe that in tumors where CXCL1 or 2 are silenced the rate of apoptosis is higher. Ok then, it is something to do with apoptosis…The authors then demonstrate that the action of CXCL1 and 2 in breast cancer cells is non-cell autonomous meaning that for CXCL1 and 2 to diminish apoptosis the immune cells of the host are needed. By means of flow cytometry they find that specifically a subpopulation of immune cells called granulocytic Myeloid Derived Suppressor Cells (MDSCs) are needed. To sum up, breast cancer cells use CXCL1 and 2 to attract some immune cells (MDSCs) that help them avoiding apoptosis.
So what is next? The mechanism; how MDSCs help breast cancer cells to avoid metastasis? The authors use an intelligent strategy to address this question; first they perform gene expression assays of whole breast tumors and they filter the results looking for secreted and membrane proteins associated with high CXCL1 or 2 expression; the rationale for this is easy, whatever MDSCs do in tumoral cells it must be via secreted proteins (paracrine way) or via cell-to-cell contacts. This gives a list of candidates. Afterwards they analyze gene expression in isolated MDSCs obtaining another set of genes. Then they cross lists looking for candidates appearing in both and…Bingo! They find two very interesting suspects, S100A8 and S100A9 (figure 2). S100A8 and A9 are calcium binding proteins that have been previously associated with chronic inflammation and cancer. The authors then use mouse chimeras in which S100A8 and S100A9 are specifically knocked out in the MDSCs population. They show that tumors grow less and there are less and smaller metastases in the absence of S100A8 and A9 in the MDSCs (figure 2). Importantly, they also relate S100A8 and A9 expression with reduced overall survival in human samples giving clinical relevance to their results (figure 2). All in a nutshell, breast tumors use CXCL1 and 2 to attract MDSCs that in turn provide S100A8 and A9 increasing the resistance to apoptosis of tumoral cells.
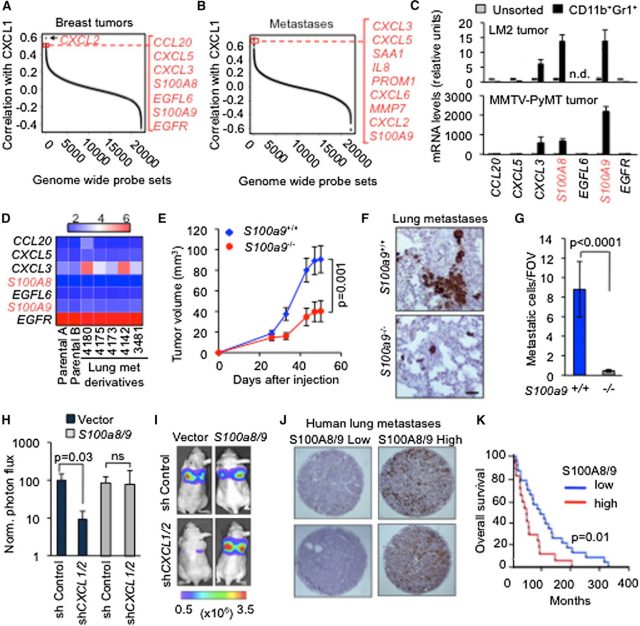
The rest of the article explains how chemotherapy stimulates this CXCL1/2-S100A8/9 axis. For every treatment used they demonstrate that successive chemotherapy rounds select for tumoral cells that express high levels of CXCL1 and 2. Importantly, this is not only a classical Darwinian process in which an evolutive pressure (chemotherapy) selects for random cancer variants with high CXCL 1 or 2 expression; in this case, chemotherapy actively induce CXCL1 and 2 expression in cancer cells. This is so because chemotherapy provokes a general secretion of pro-inflammatory mediators, including TNF, in the patients. TNF in turn increases CXCL1 and CXCL2 expression in tumoral cells via NF-b pathway paving the way for MDSCs recruitment, apoptosis resistance and metastasis. Finally, to add translational impact to their findings, the authors combine standard chemotherapy and CXCL1 and 2 inhibitors to show that this dual treatment is much more effective than chemotherapy alone.
“If you know the enemy and know yourself, you need not to fear the results of a hundred battles”. Master strategist Sun Tzu wrote this some 25 centuries ago. Cancer is such a threat because it is both an adversary and part of our own nature…But, little by little, we are learning and dismounting the stratagems of this formidable enemy within.
References
- Murphy K, Travers P, Walport M: Janeway´s Immunobiology, Seventh edn: Garland Science; 2008. ↩
- Balkwill F, Mantovani A: Inflammation and cancer: back to Virchow? Lancet 2001, a(9255):539-545. ↩
- Acharyya S, Oskarsson T, Vanharanta S, Malladi S, Kim J, Morris PG, Manova-Todorova K, Leversha M, Hogg N, Seshan VE et al: A CXCL1 paracrine network links cancer chemoresistance and metastasis. Cell 2012, a(1):165-178. ↩
4 comments
Felicidades!!!….enhorabuena!!!….¡¡¡lo tienes merecido!!!. Me lo he leído todo (me he enterado perfectamente) y me parecen estupendos los comentarios sobre ti, Manuel Tardáguila.
Fuertes abrazos y mi apoyo incondicional.
Ahora que traduzcan esto…jejejejeje
Me siento muy honrada de conocer a Manuel Tardáguila y el maravilloso trabajo de investigación que realiza. Apoyemos la INVESTIGACIÓN.
Carmen Lopez
Congratulations for a fabulous information article. We are in the middle of hopeful discoveries… Society needs to push harder in this field in order to find the whys and the hows!!!!
Magnifico Manuel y muy interesante. No sabía el modo en que las células cancerígenas se comportan y esto ha sido muy educativo. Y más aun viniendo de alguien que conozco y que tiene una vis cómica que me encanta. Bravo