Mysterious vaults and neuronal regeneration
From a certain point of view, eukaryotic cells could be described as microscopic power plants equipped with factories and energy-generating platforms, inside of which highways and compartments are constantly built and rebuilt, and a fluent traffic provides a constant recycling of structures while getting rid of garbage. The trafficking also involves material coming in and out of the nucleus, the command center. This vision of the cells is constantly being enriched by new discoveries, although nowadays we tend to think that there is nothing new on the horizon regarding cellular structures and organelles. However, recent findings still challenge our imagination: some oddities remain unknown within the plasmatic walls of living cells, and among these, we find the amazing and huge protein structures known as vaults.
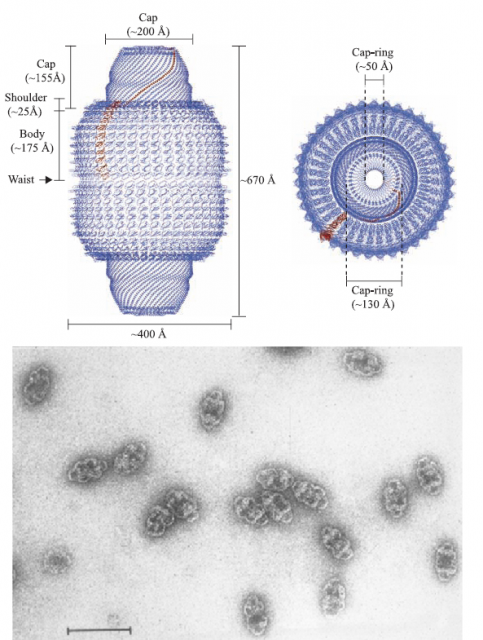
Vaults are macromolecular complexes constituted by a conglomerate of repetitions of a small protein, which in turn generates a barrel-shaped structure with a hollow interior. The overall structure is bigger than two ribosomes together… and that is really big for a cell (Figure 1). Biologists have been intrigued more than twenty years by these macrostructures, equipped with their own set of small RNA particles and a few more proteins, mostly related to RNA processing (this features are striking since they link vaults to some types of RNA viruses). Since the determination of the crystallographic structure of the whole complex was achieved 1, a possible role of vaults as carrier complexes for other particles, proteins or nucleic acids, was suggested; no clear data has confirmed this hypothesis, and vaults (either full or empty, who knows) are found largely in the cytoplasm and in a lesser proportion, associated to the nuclear envelope.
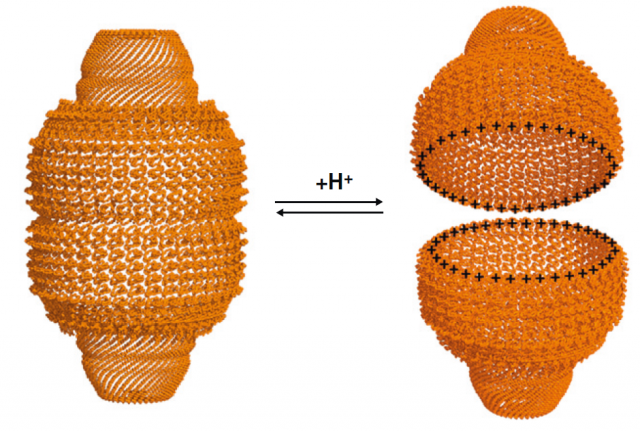
Many independent reports have linked vaults, or more precisely their predominantly constituent protein called Major Vault Protein (MVP; nothing to do with sport players), to cellular processes that involve signal transduction pathways mediated by Mitogen-activated protein kinaes (MAPKs), or the tumor suppressor protein PTEN. Is MVP a scaffolding protein that interacts with other partners besides forming the huge vaults? Are vaults carrier platforms that deliver the goods from one point of the cytoplasm to the surroundings of the nucleus? The opening mechanisms of the vault (they have been shown to spontaneously open and re-arrange again in a pH-dependent manner) 2 (Figure 2) and more reports linking them to lysosomes will probably generate more interesting questions. But today, we will focus in a brand new proposal of an intriguing role for these little giants.

The recent work by Pan and collaborators 3 suggests novel implications for the MVP. In this case, the focus of the study is the process of regeneration in zebrafish, a popular animal model in research (Figure 3). Among many other interesting advantages, zebrafish show a capacity of recovering central nervous system function after severe injury, regenerating axons and creating new connections between neurons in a way that most mammals can only dream of. For this reason the researchers decided to dig deeper into this amazing regeneration capacity, and analyzed the pattern of expression of genes seemingly involved in the process. And there, in an unexpected scenario, appeared the vaults.
The reader may think that vaults are a recent invention of complex organisms, a refined structure strictly present in “superior” animals. Well, here’s another slap in the face for anthropocentrism: these incredible particles are evolutionarily conserved and broadly extended in all eukaryots, from unicellular yeasts to the more complicated vertebrates [4]. Many organisms possess vaults or strikingly similar structures, what reinforces the idea of a functional relevance for the processes mediated by them.
As we said earlier, vaults are constituted in a vast majority by the small protein named MVP. The increase in its expression levels, at a certain time since generating neural injury, suggested a role for vaults in the process of regenerating axons; besides, co-localization with other important proteins in neuronal growth (i.e. nestin, a filamentous protein implicated in the growing process of axons), seemed to confirm this hypothesis. After knockdown of MVP expression in the particular region of the spinal cord, where it had been found to be enhanced, the fish were not able to recover mobility as did other injured fish with a normal MVP expression. Furthermore, the MVP-Knockdown neurons also suffered a reduction in the number of synapses.
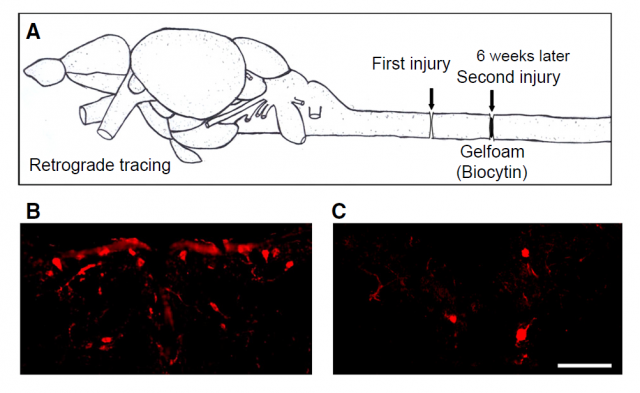
The conclusions of the work are obvious: MVP is present in the correct place at the correct moment, and its elimination correlates with a decrease in the capacity of zebrafish to regenerate its neuronal populations after a severe spinal cord injury (Figure 4), reducing the number of synapses and impeding locomotor recovery. How this small protein which generally forms part of a huge cellular structure, remains elusive to the authors. They suggest that the same evidence that links vaults to cell survival and proliferation, involving molecular pathways regulated by other signaling modules, may have something to do with the effects seen in neuronal proliferation and regeneration in zebrafish.
But the main, deeper questions remain unanswered: in which ways do vaults perform their tasks? How do they distinguish between a particular mission and others? Is the transport of molecules the key of their function? Works like the one discussed in this article surely show a clear implication of vaults in concrete and critical cellular processes; how they do it, is still a mystery.
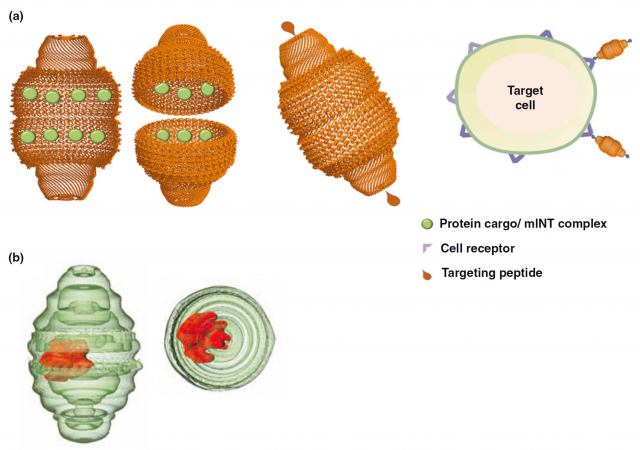
The finding of vaults participating in the central nervous system is encouraging, since the conservation of the protein in humans makes us hope a similar role that could constitute a focus of research for neuronal regeneration or, at least, healing of injured neurons. But the most promising issue concerning vaults is that, impatient as only human beings can be, we have not waited for the complete unveiling of their mysteries: since the discovery of these structures and the particular properties found in a first instance, many groups have been working in the use of these giants of the cell as carrier structures that could be helpful for drug delivery and treatment of diverse pathologies 4. There have already been attempts of engineering vaults attaching particular antibodies that target them to cancer cells, or filling vaults with vaccines without the need of adjuvants (Figure 5).
We may be assisting to a new era for transfection strategies, encapsulation research, or even in vivo therapies that get closer than ever to the eagerly expected genetic therapy that still remains elusive. In any case, there are yet many things to discover about these fascinating structures, their origin, their physiological functions, and hopefully other biotechnological applications which we cannot even imagine. Not bad for a big hollow device that seemingly wanders around through the cytoplasm, waiting for someone to discover its real purpose in life.
References
- Tanaka H, Kato K, Yamashita E, Sumizawa T, Zhou Y, et al. (2009) The structure of rat liver vault at 3.5 angstrom resolution. Science 323: 384-388. ↩
- Querol-Audi J, Casanas A, Uson I, Luque D, Caston JR, et al. (2009) The mechanism of vault opening from the high resolution structure of the N-terminal repeats of MVP. EMBO J 28: 3450-3457. ↩
- Pan HC, Lin JF, Ma LP, Shen YQ, Schachner M (2013) Major vault protein promotes locomotor recovery and regeneration after spinal cord injury in adult zebrafish. Eur J Neurosci 37: 203-211. ↩
- Casanas A, Guerra P, Fita I, Verdaguer N (2012) Vault particles: a new generation of delivery nanodevices. Curr Opin Biotechnol 23: 972-977. ↩
3 comments
Interesting article, and well written, thanks!
FYI Vaults are absent in all fungi and are not found in yeast.
Thank you very much! And thank you also for the correction, it’s true that the absence in yeast is another striking feature of the vaults, I don’t know in which moment I forgot about this fact, or what I read that made me make that mistake. I’ll get it corrected.
I just came back to the original article and at some point it says “MVP accounts for up 75% of the total complex and is highly conserved from mammals to fungi”; probably this is what confused me…