Born to be a mother, grown to be wild
Born to be a mother, grown to be wild
Authors:
Ana Martín-Sánchez is currently pursuing her PhD at the Department of Functional Biology and Physical Anthropology of the University of Valencia, Spain
Carmen Agustín-Pavón is a lecturer and researcher at the Functional Neuroanatomy group of the Department of Medicine of the Jaume I University, Spain
Motherhood is a vital and challenging job for most mammalian females: the survival and welfare of the offspring mostly depends on maternal care. The quality of maternal care has a huge impact on the development of the young. In humans, those children suffering from low parental bonding –cold, distant parent-child relationships- might be at higher risk of depression and anxiety during adulthood. Thus, investigating how mother-infant interaction is established is essential to understand why and how this bonding fails in some mothers and their children. Laboratory animals such as mice have helped us a lot in the investigation of the neural substrate of maternal care. Thanks to them, we understand most of the hormonal and physiological changes that are key for the display of maternal care, and researchers have mapped the brain nuclei that control this behaviour.
However, there is an aspect of maternal behaviour that is far less understood than maternal care: maternal aggression. During late pregnancy and the beginning of lactation, new mothers become very aggressive and fiercely attack intruders in the proximity of the nest. This is an adaptive behaviour that helps the mother protecting her offspring from potentially infanticide males, who usually kill unfamiliar pups. In fact, mouse mothers are much more aggressive against males than they are against other females or castrated males. By contrast, female mice that are not breeding are docile and rarely display aggressive behaviour against conspecifics.
Interestingly, virgin female mice seem wired for maternal care, but not for aggression. In our latest paper1, we have demonstrated that when virgin albino mice are housed with their pregnant sisters, they act as a kind of godmothers from the moment that the pups are born. The godmothers are as quick as their mom sisters in retrieving and grouping the pups in the nest, they lick and groom them as much as mommies do and, even if they cannot nurse them, godmothers crouch over the pups at higher frequency than the mums.
The fact that maternal care is rapidly induced in virgin laboratory mice by contact with foster pups is not a new discovery, other labs had showed this by using different protocols. What it’s new in our study is that we also tested whether godmothers would attack intruders. And we showed that they don’t: godmothers seem born to be mothers, not to be wild. Instead, godmothers display friendly socio-sexual investigation of the male intruders, i.e. sniffing the face and the anogenital region of the males –yet some godmothers might seldom show one or two bouts of short attack. Thus, the physiological changes occurring during pregnancy and lactation might switch on some structures in the brain of the new moms that enable the aggressive state.
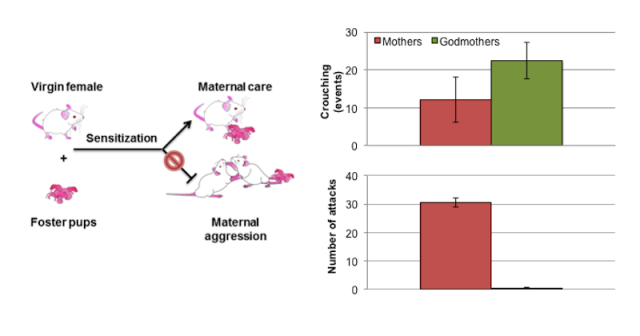
Now we want to use our mothers and godmothers to investigate which brain nuclei and neurotransmitters are involved in the switching on of the aggressive state. We already have several candidates in the pipeline. For example, we have shown that maternal aggression is displayed only in the mother’s home cage, suggesting that the hippocampus, involved in spatial recognition, might modulate maternal aggression.
Further, we have shown that a most efficient trigger of moms’ rage is a male pheromone –a chemical signal detected by the olfactory systems that has communicative purposes. This pheromone is darcin, a component of the male urine identified by Jane Hurst, Rob Beynon and colleagues in the Institute of Integrative Biology in the University of Liverpool –and christened after Jane Austen’s male character in Pride and Prejudice, the attractive Mr Darcy. Indeed, the Liverpool researchers demonstrated that darcin is attractive and induces contextual learning in virgin females looking for potential partners2. In collaboration with Hurst and Beynon’s lab, our lab demonstrated that darcin is not very attractive for mothers, but rather it elicits maternal aggression3. The following videos show how a mouse mom interacts more or less peacefully with a castrated male, whereas she attacks ferociously a castrated male that was sprayed with darcin. This shows how the presence of a single pheromone profoundly affects a mouse’s behaviour.
Video 1. A mouse mom (at the upper-left corner at the beginning of the video) interacts with a castrated male: she sniffs him and little else.
Video 2. A mouse mom (at the right handside of the cage at the beginning of the video) attacks a castrated male sprayed with the pheromone darcin. Darcin is one of the pheromones that signals maleness, and thus it means for mothers that the carrier is a potential pup killer. Note how the tail of the female rattles as she prepares the attacks, and the attempts of biting directed to the male snout.
These data suggest that brain nuclei related to olfaction, including some of the areas of the amygdala –a core region of the emotional brain- that are profusely interconnected with the olfactory system, are likely to play a critical role in maternal aggression.
Motherhood might thus induce plastic changes in those mentioned brain regions, and those changes will make the peaceful female mouse brain turn to belligerence. Although it might seem counterintuitive, investigating how maternity tunes the female brain for selective aggression might provide new clues about how unselective violence arises. Violence is a present-day problem in our society, and we hope that our future research in our mouse mothers and godmothers will help devising better strategies to combat it.
Ana Martín-Sánchez @nosoyunbicho, PhD student at the University of Valencia
Carmen Agustín Pavón @CarmenAgustin, Researcher and Lecturer at the University Jaume I of Castelló
References
- Ana Martín-Sánchez, Guillermo Valera-Marín, Adoración Hernández-Martínez, Enrique Lanuza, Fernando Martínez-García & Carmen Agustín-Pavón. (2015) Wired for motherhood: induction of maternal care but not maternal aggression in virgin female CD1 mice. Front. Behav. Neurosci. doi: 10.3389/fnbeh.2015.00197 ↩
- Roberts SA, Davidson AJ, McLean L, Beynon RJ, Hurst JL. (2012) Pheromonal induction of spatial learning in mice. Science 338(6113):1462-5. doi: 10.1126/science.1225638 ↩
- Martín-Sánchez A, McLean L, Beynon RJ, Hurst JL, Ayala G, Lanuza E, Martínez-Garcia F. From sexual attraction to maternal aggression: when pheromones change their behavioural significance (2015) Horm Behav. 68:65-76. doi: 10.1016/j.yhbeh.2014.08.007 ↩
1 comment
[…] Otro aspecto básico en la evolución de las especies de mamíferos es el cuidado que las madres (y algunas veces los padres) prestan a sus crías durante periodos más o menos prolongados de tiempo. Y asociado a este comportamiento […]