Neural precursors accelerate the development of tumors
Neural precursors accelerate the development of tumors
Author: José R. Pineda got his Ph.D. from University of Barcelona in 2006. Since 2007 he has worked for Institut Curie and The French Alternative Energies and Atomic Energy Commission. Currently he is a «Ramón y Cajal» researcher affiliated to UPV/EHU and the Achucarro Basque Center for Neuroscience. He investigates the role of stem cells in physiologic and pathologic conditions.
“The brain is considered an isolated tissue from the rest of the body thanks to the blood-brain-barrier, which separates it at cellular level with a very strict control”. That was supposed in the past, at least until now… In 2019 a striking discovery led by the researcher Moreno-Jimenez demonstrated human neurogenesis is abundant and persists during adult life in healthy subjects 1. However, a bigger discovery was also announced with no media impact. Nobody could imagine that neural precursors which their unique role is to sustain brain neurogenesis, are also able to exit from the brain and migrate towards developing tumors to boost tumor growth and kill us faster. That could be the summary of the work led by Philippe Maupffrey2 and collaborators at The French Alternative Energies and Atomic Energy Commission in an impressive demonstration published in the journal Nature.
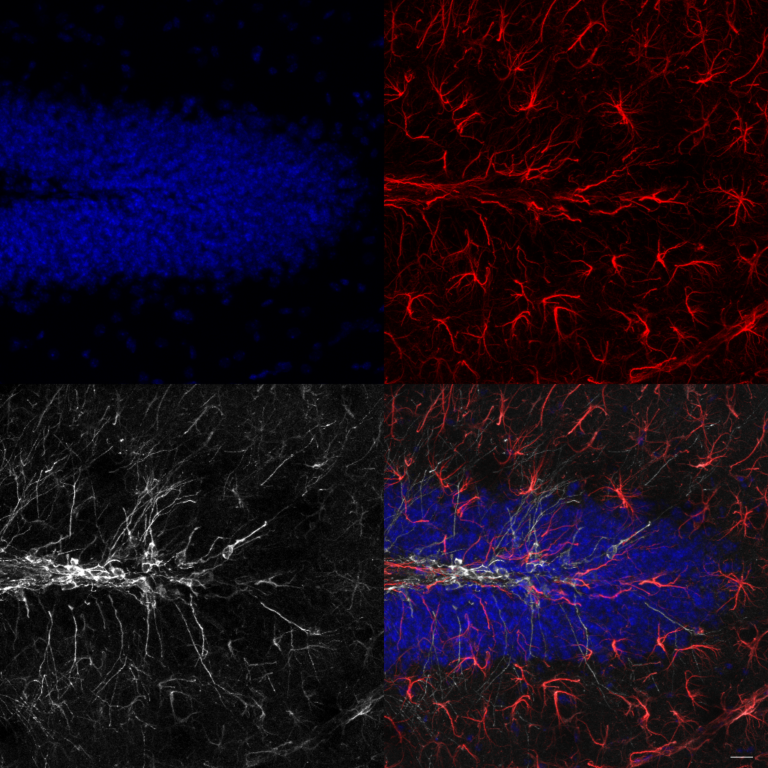
The work led by Philippe Maupffrey 2 uses a mouse model that express MYC transgene (an oncogene involved in the regulation and increased expression of many genes, some of which are involved in cell proliferation, contributing to the formation of cancer) in the murine prostate. In other words, they produced prostate cancer (the prostate is very far from the brain). In order to be very strict, they decided to control the neurogenesis, or at least, the production of immature neural progenitors (DCX+). For this purpose, through a genetic engineering technique, they elegantly were able to “turn on” the production of DCX+ cells at will. Furthermore, in order to track these newly generated DCX cells were engineered to express a green fluorescent protein, becoming unequivocally tagged. They find out that newly produced fluorescent DCX+ cells were found in prostate tumors but not in healthy prostate tissues from littermates without the MYC transgene. Interestingly (and hopefully!) fluorescent DCX+ cells were negative for stem cell markers although isolated cells cultured in vitro maintained the capabilities to proliferate and differentiate into neurons. These observations raised the question about its role in vivo. Histological analysis at different time-points of mice bearing the prostate tumors demonstrated that DCX+ cells again were found in the tumors but not in the surrounding healthy tissue. Moreover, they found over time a production of nerve fibers fluorescently tagged, demonstrating that these produced nerves came from the DCX+ neural precursors.
With this appealing finding, the next question for a neuroscientist was to discover which type of nerve is produced in order to gain knowledge about its role and further implications. The authors determined that this nerve signaling was adrenergic, a type of nerve previously known to contribute to prostate cancer progressiom 3 . In order to corroborate the origin of DCX+ cells in the tumor, the authors used a red-colored reporter by lentiviral infection placing the virus in a neurogenic region of the murine brain called subventricular region (SVZ). Using this strategy, if red colored cells were found at the tumor, the scientists will know that these cells came from the unique red-colored zone in the brain. By this way, the authors demonstrated that DCX+ cells (able to innervate the prostate tumor) egressed from SVZ (neurogenic) region of the brain. Surprisingly, they found that this “cellular escape” from the brain only happened in cancerous mouse model but not in healthy littermates.
As neuroscientists, we know that in the brain there is another neurogenic region, more relevant for the humans that is located in the hippocampi (brain regions involved in the circuitry of memory) known as dentate gyrus (DG)1. Conscious of the big implications of their finding, the authors repeated the experiment labeling all the DG cells using again the red-colored reporter by lentiviral injection. Surprisingly (and hopefully!), this time no cells carrying red signal were found in the tumor. These results suggested the existence of difference vascular permeability between both neurogenic regions. Only the neural progenitors of the SVZ were able to penetrate and cross the blood–brain barrier and migrate away.
Looking at these findings restrospectively, we may ask if this phenomenon is specific of prostate cancer or it may be a general feature irrespective of the type of tumor. The answer of this interesting question was provided by the authors repeating the paradigm in another mouse model bearing breast cancer. They again found the DCX+ cells in breast tumors!!!
From an oncological point of view, the real risk of suffering a cancer is not only the stage of tumor development, but the risk of tumor metastasis. Metastasis is the pathogenic spread from an initial or primary site to a different or secondary (tertiary etc.) sites within the host’s body. That leads to an uncontrolled dissemination limiting the effectiveness of surgical resection and causing future tumor recurrence. For this reason, to check the real implications of tumor innervation from precursors of the neurogenic niche, the authors decided to induce tumor metastasis. They made xenografts of cancer cells. Using this approximation, cancer cells disseminates very fast spreading into different host tissues such as colon, liver, lungs… After histological analysis they found again DCX+ neuronal precursors infiltrated in xenograft and metastatic tissues. With this experimental design, the authors demonstrated that these events are a general feature of the development of cancer.
Last but not least, to prove the implication of DCX+ neuronal precursors in the tumor development, the authors decided to repeat the experiment modifying the DCX+ cells with an inducible diphtheria toxin receptor. This smart strategy allowed choosing to kill all DCX+ at the desired moment to prove the consequences of DCX+ cell elimination during the growth of tumor. They found that the loss of DCX+ cells significantly inhibited the engraftment of metastatic tumor cells, suggesting that DCX+ neurogenic cells played a critical role for the early staged of tumor development. To fully corroborate this asseveration, additional experiments grafting DCX+ positive cells into tumors resulted in an increase of tumor growth several weeks after transplantation.
In conclusion, the work of Maupffrey and collaborators unravels the origin of the nerves that surrounds tumor cell mass (a process known as perineural invasion). Moreover, it is known that human SVZ is a quiescent neurogenic niche that may reactivate under pathological conditions (such as strokes). These results suggest that during tumor development, SVZ may wake up and to be hickjaked to provide neural progenitors that initiate neurogenesis during tumor formation. Further studies are necessary to characterize the signaling events responsible for the egression of neural progenitor cells from the brain towards the tumors and the mechanisms to prevent it.
References
- Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, Rábano A, Cafini F, Pallas-Bazarra N, Ávila J, Llorens-Martín M. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019 Apr; 25(4):554-560. doi: 10.1038/s41591-019-0375-9. ↩
- Mauffrey P, Tchitchek N, Barroca V, Bemelmans A, Firlej V, Allory Y, Roméo PH, Magnon C. Progenitors from the central nervous system drive neurogenesis in cancer. Nature. 2019 May;569(7758):672-678. doi: 10.1038/s41586-019-1219-y. ↩
- Magnon C, Hall SJ, Lin J, Xue X, Gerber L, Freedland SJ, Frenette PS. Autonomic nerve development contributes to prostate cancer progression. Science. 2013 Jul 12;341(6142):1236361. PMID: 23846904 doi: 10.1126/science.1236361. ↩