Zombosomes: The rise of astrocytic couriers in the propagation of brain disease
Zombosomes: The rise of astrocytic couriers in the propagation of brain disease
Author: José R. Pineda earned his Ph.D. from the University of Barcelona in 2006. Since 2007, he has conducted research at Institut Curie and the French Alternative Energies and Atomic Energy Commission. He is currently a researcher at the University of the Basque Country – EHU, where his work focuses on the role of stem cells in both physiological and pathological contexts.
During the last decade, neurodegenerative diseases have often been described as fires spreading silently from one neuron to another. We knew that misfolded proteins such as alpha‑synuclein could travel across neural circuits, igniting new pathological hotspots along the way. What we did not know is that, behind the scenes, astrocytes —those tireless caretakers of the central nervous system— were capable of shedding small “living couriers” without a nucleus, capable of moving on their own and carrying pathological cargo. That is the essence of the work presented by Dakhel and collaborators: the discovery of an unexpected type of intercellular messenger known as zombosomes 1.
The next question was whether such strange entities could have a role in the context of disease. The authors tried to determine if alpha‑synuclein pre‑formed fibrils (PFFs), a well‑established model for Parkinson’s‑like pathology could be present in zombosomes. Because astrocytes readily internalize these fibrils but fail to degrade them completely, the researchers observed zombosomes budding off that contain fluorescently labeled alpha‑synuclein aggregates. These migrating zombosomes were then taken up by neighboring astrocytes and, crucially, by neurons!! In exposed neuronal cultures, they induced the appearance of phosphorylated alpha‑synuclein deposits (precisely the pathological signature seen in affected human brains!!). Even more striking, when these zombosomes were delivered to human cortical organoids, they infiltrated deep layers of the tissue and triggered robust alpha‑synuclein pathology, increasing both soluble and insoluble fractions of the protein. Despite delivering far less total protein than whole cells, zombosomes appear more efficient at spreading pathology, hinting at a uniquely potent mechanism of cargo delivery.
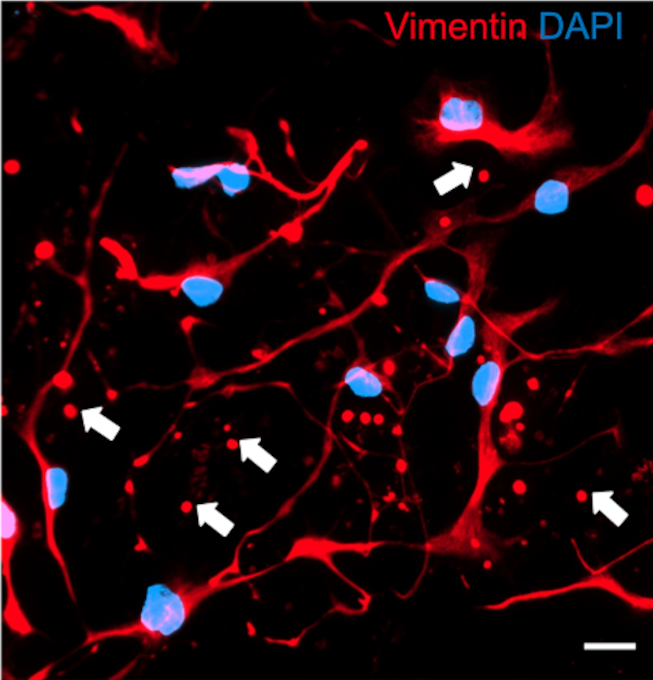
But the story does not end in vitro. When researchers examined human post‑mortem brain tissue, they also identified vimentin‑rich, nucleus‑free spherical structures scattered throughout the striatum (Nuclei Caudate and Putamen) as well as in the substantia nigra, both regions deeply involved in the progression of Parkinson’s disease. These bodies matched the size and morphology of cultured zombosomes and, importantly, co‑localized with phosphorylated alpha‑synuclein through “PLA-Assays” (PLA means “proximity ligation assay” and emits a fluorescent signal of molecules very close in the space). Interestingly, their numbers do not differ significantly between Parkinson’s and control brains, suggesting that zombosomes may exist under normal physiological conditions, becoming pathological only when hijacked by disease‑related cargo.
To place this discovery into the broader landscape of glial biology, it helps to remember that astrocytes are the true responsibles of proper brain function. They help the neurons by orchestrating energy allocation, regulating neurotransmission, clearing the debris, remodelling the synapses, and even become the intermediaries between the neurons and the vasculature. Such versatility also brings vulnerability: the very pathways designed for homeostasis can be repurposed by toxic proteins. We already knew of nanotubes, tunneling structures, and extracellular vesicles as channels for this kind of hijacking. Zombosomes now add a new fast‑track route, one endowed with autonomous motility, firm substrate adhesion, and the ability to carry both organelles and aggregated proteins. If an exosome is a sealed letter, a zombosome feels more like a motorcycle courier equipped with tools and fuel. Indeed, comparisons with other anucleated structures clarify what zombosomes are—and what they are not. They are not platelets, which originate from megakaryocytes and lack vimentin; they are not migrasomes, which do not move independently; and although they resemble exophers in size and content, zombosomes differ fundamentally in their glial origin and inherent motility. Even recently described “blebbisomes” from cancer cells, despite sharing some features, come from distinct biological contexts. What emerges is a broader and more diverse landscape of anucleated spheres in biology, within which zombosomes occupy a newly defined niche.
However, no elegant discovery comes without caveats. Most of the functional demonstrations occur in culture systems or organoids. Observing zombosome motility and cargo transfer in vivo, within the densely packed architecture of an intact mammalian brain, remains an outstanding challenge. Identifying selective zombosome markers will help track them more precisely, and characterizing the regulatory pathways that trigger their formation will determine whether they represent a stress response, a homeostatic tool, or both. The authors themselves stress that the next major step will be validating these findings in living organisms and exploring how zombosomes behave in disease models that replicate Parkinson’s pathology more faithfully. Still, the conceptual leap offered by the work of Dakhel and collaborators published in the prestigious Cell Journal is remarkable. If the brain possesses a glial‑derived system of motile couriers capable of transporting organelles and aggregated proteins, then both healthy and pathological communication must be reconsidered. Under normal conditions, zombosomes might redistribute resources or signals across short distances. Under pathological pressure, they could inadvertently disseminate harmful seeds, accelerating disease progression. Understanding when a zombosome becomes an ally and when it becomes an unwitting accomplice will be essential for future therapies. Could their formation be inhibited when astrocytes are overwhelmed by proteotoxic stress? Could their cargo be neutralized before delivery? Or—turning the concept on its head—could zombosomes be harnessed as natural delivery vehicles for therapeutic molecules or healthy mitochondria? These are the kinds of translational questions that emerge from a discovery that reshapes our map of neural communication.
There is also a human element woven into the narrative of this study. Astrocytes are not villains, but versatile responders trying to cope with stress. Their attempt to manage overwhelming loads of aggregated protein—by repackaging and exporting material—may lead to collateral damage. Zombosomes may be part of that ambiguous response: improvised couriers meant to redistribute burdens that, when carrying toxic cargo, inadvertently pass the problem along. Biology, as always, deals more with nuance than with intent. Looking ahead, zombosomes may become valuable biomarkers or therapeutic targets. If their proteomic signature can be distinguished clearly and their abundance tracked across disease stages, they could offer new insights into neurodegenerative progression. Conversely, intercepting their formation or blocking their infiltration into vulnerable tissues might slow the spread of pathology. The tools to do so are still in development, but the conceptual groundwork is already here. In this sense, the study does more than describe an unusual phenomenon—it reframes our understanding of how cells communicate in health and disease.
If science teaches us anything, it is that perspective changes often arise from looking again at what was always in front of us. Astrocytes have been present in every histological slice since the dawn of neuroscience. Now, from that familiar lineage emerges an unexpected system of motile, organelle‑bearing shuttles that may shape how neurodegenerative diseases advance. The name “zombosomes” will certainly capture attention, but what may endure is the profound shift in how we understand glial communication—and the new therapeutic horizons it may open.
References
- Abdulkhalek Dakhel, Chiara Beretta, Tobias Mothes, Sona Hakhverdyan, Wojciech Michno, Jinar Rostami, Anna Erlandsson. Zombosomes are anucleated cell couriers that spread α‑synuclein pathology. Cell Reports 45, 116831 (2026) doi: 10.1016/j.celrep.2025.116831 ↩