Molecular origami: Crafting ultrasmall nanogels through intramolecular architecture
Molecular origami: Crafting ultrasmall nanogels through intramolecular architecture
Nanogels are among the most interesting examples of how chemists can build useful structures by working at the scale of single molecules. They are tiny soft particles made from polymers, long chain-like molecules, that are linked together into small three-dimensional networks. Because they can hold water and trap other molecules inside, nanogels are especially attractive for medicine, sensing, and the controlled delivery of small compounds. While most nanogels are much larger than individual polymer molecules, often tens or even hundreds of nanometres across, an even more elegant idea is to create a nanogel by folding and linking just one polymer molecule into itself, rather than joining many molecules together.
This produces an ultrasmall object, still soft and functional, but much more uniform and precisely controlled. A recent advance 1 explores exactly this strategy using polyglycerol, a highly branched polymer well known for being biocompatible and rich in hydroxyl groups. These hydroxyl groups are chemically useful because they can form reversible bonds with boronic acids. Instead of using straight polymer chains, the researchers worked with branched and hyperbranched cyclic polyglycerols. These are compact, loop-like molecules that resemble tiny molecular trees without “loose ends.” Some were relatively small, while others were much larger and more densely branched, a difference in architecture that turned out to be crucial.
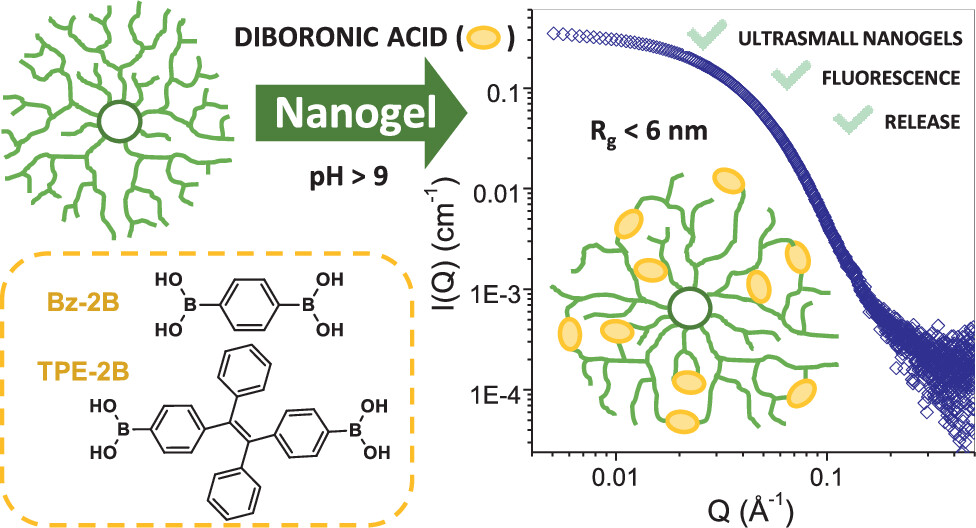
The goal was to gently “tie” parts of each molecule together from the inside. To do this, diboronic acids were added under mildly basic and highly dilute conditions. Boronic acids can react with neighbouring diol groups (pairs of nearby alcohol groups) to form boronic esters, which are dynamic covalent bonds. “Dynamic” means the bonds are reversible: they can form, break, and reform depending on conditions such as pH. This allows the structure to reorganize rather than becoming permanently locked too early. Under these specific conditions, the diboronic acids mostly connected different parts of the same polymer molecule rather than linking separate molecules together.
The result was an intramolecularly cross-linked nanogel. These nanogels were extremely small, with radii of gyration below 6 nanometres, making them closer in size to large proteins, like hemoglobin, than to conventional polymer particles. Importantly, their overall dimensions changed very little after cross-linking. This may sound surprising, but it is actually strong evidence that the reaction happened inside individual molecules; if many molecules had linked together, the particles would have become much larger. Small-angle X-ray scattering confirmed that aggregation was minimal and that the process mainly produced single-molecule nanogels rather than clusters.
The internal shape of these structures depended strongly on molecular weight. The smaller branched polymers behaved more like open, loose structures with an irregular, fractal-like character. In contrast, the larger hyperbranched polymers formed compact, almost spherical objects that looked much more like true nanoparticles. This shows an important principle in macromolecular chemistry: the same chemical building blocks can behave very differently depending on architecture. Shape determines function.
To understand what was happening inside these networks, the researchers used a second diboronic acid containing a tetraphenylethylene (TPE) unit. This molecule acts as a “glowing” bridge that becomes strongly fluorescent only when its internal rotations are restricted. In free solution, it glows weakly, but when trapped and locked as a cross-linker in a confined environment, it lights up brightly. This made TPE a clever molecular probe. In the smaller polymers, TPE was found mainly near the outer regions where movement was less restricted. In the larger hyperbranched nanogels, however, the TPE bridge became trapped deeper inside, where its motion was strongly limited. As a result, fluorescence increased dramatically, up to fifty times stronger than the free molecule, allowing researchers to “see” the density of the nanogel’s interior.
Finally, the study tested whether these ultrasmall nanogels could hold guest molecules. Using rhodamine B, a fluorescent dye, as a model, the researchers found the nanogels could physically trap the dye and release it gradually over time. This release depended on how densely the structure was cross-linked and how large the original polymer was. More compact and strongly cross-linked systems held the dye more effectively. Controlled release is a primary reason nanogels are attractive for biomedical use. A carrier that protects a molecule and releases it slowly, or under specific pH conditions, is invaluable for drug delivery.
This research connects structure, function, and molecular design directly. By changing polymer size and branching, compactness can be controlled; by choosing different diboronic acids, fluorescence and guest retention can be tuned. Because the linking chemistry is reversible, the system remains adaptable rather than rigid, showing how modern polymer chemistry is moving toward precise, responsive nanoscale objects.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may have been copied verbatim or almost verbatim from the referenced research paper/s.
References
- E. Gomez Urreizti, P. Malo de Molina, C. A. Pagnacco, J. A. Pomposo, R. Lund, and F. Barroso-Bujans (2026) Ultrasmall nanogels from intramolecular cross-linking of (hyper)branched cyclic polyglycerols with diboronic acids Macromolecules doi: 10.1021/acs.macromol.5c02780 ↩