Unveiling the origin of the record superconductivity
Unveiling the origin of the record superconductivity
Achieving room temperature superconductivity is among the most pursued but elusive goals of scientists. A paper uploaded to the arXiv 1 in December 2014 claims to have observed superconductivity as high as 190 K in hydrogen sulfide at high pressure, breaking all the records thus far. If this observation is confirmed, cuprates will be knocked from their position as the highest temperature superconductors. The result represents a huge step towards a room-temperature superconductor and remarks that very high transition temperatures can be achieved with simple compounds.
![Fig. 1: High-pressure experiments, as the one that has observed the record superconductivity [1], are normally perfomed making use of diamond anvil cells. Credit: http://www.earth.northwestern.edu/research/jacobsen/](https://mappingignorance.org/app/uploads/2015/04/Jacobsen-Diamond-Anvil-Cell.jpg)
Hydrogen sulfide (H2S) at ambient conditions is a gas recognizable for its rotten-egg odor. Despite its chemical similarity to water (H2O), it is poisonous, flammable, and explosive. H2S is present in nature as it might result from the bacterial breakdown of organic matter when oxygen lacks, and occurs in volcanic as well as natural gases. However, under pressure its properties completely change. It first becomes a molecular insulating solid, then the molecules dissociate forming an atomic solid, and, finally, it becomes a metal around 96 GPa 2. Now, the experiments performed using a diamond anvil cell (see Figure 1) by Drozdov et al. indicate that compressing hydrogen sulfide further, up to 200 GPa, it can superconduct at 190 K, the highest superconducting critical temperature (Tc) ever observed [1].
The observation is revolutionary because it confirms experimentally for the first time the theoretical intuition by Ashcroft that high-temperature superconductivity could be attained in metallic hydrogen or hydrogen-rich materials 3,4. The expected extraordinary values are not originated from an exotic coupling mechanism as in the pnictides or cuprates, but from the well-understood interaction between the electrons and the vibrations of the atoms in the solid, the phonons. As it was already described in a previous post in MappingIgnorance, Tc in an electron-phonon superconductor is enhanced when the average phonon frequency and the electron-phonon coupling constant are large. According to Ashcroft, this may be the case in metallic hydrogen and hydrogen-rich compounds.
As soon as the results of the record Tc were communicated to the scientific community, many theoretical researchers devoted themselves to understand what had happened in the experiment. Some authors 5 have published theoretical indications that H2S might have dissociated into H3S and S, and have argued that superconductivity, driven by the electron-phonon mechanism, might have been observed in the H3S compound. Interestingly, before the experiments were communicated, H3S had been predicted to be a high-temperature superconductor 6. Other authors have instead claimed that the mechanism behind superconductivity is not the electron-phonon mechanism 7.
A group of scientists from the Donostia International Physics Center (DIPC) in collaboration with researchers from France, UK, Canada, and China have now shown making use of theoretical first-principles calculations that, indeed, the electron-phonon coupling mechanism explains the observed superconducting Tc, but only if anharmonic effects are considered in the hydrogen motion. The authors have published their conclusions in Physical Review Letters 8.
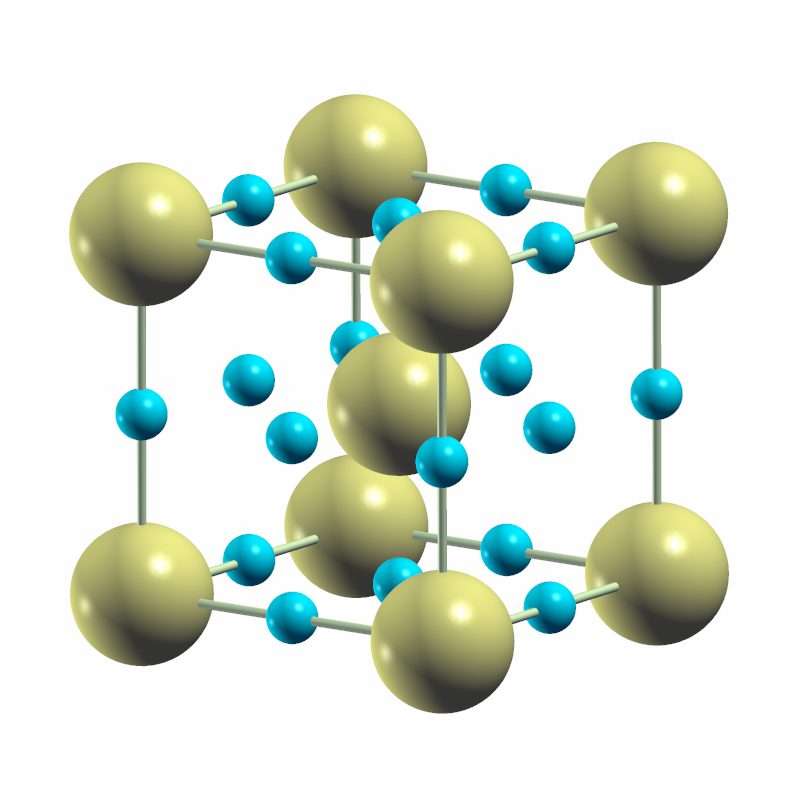
In the first place, the researchers of the latter work have studied the stability and possible decomposition of hydrogen sulfide when it is pressurized to 200 GPa. Their theoretical results indicate that H2S is not a stable stoichiometry at the pressures where the extraordinary value of Tc was observed. In fact, it can dissociate following two different mechanisms: 3H2S → 2H3S + S, which had already been described before [5], or 5H2S → 3H3S + HS2, not identified earlier. Thus, superconductivity could have occurred both in HS2 or H3S. When studying the possibility of electron-phonon superconductivity in the HS2 compound, the authors estimated that it stops superconducting approximately at 30 K, and thus cannot explain the experiment. On the contrary, the H3S phase that crystallizes in the cubic structure shown in Figure 2, has a very large electron-phonon coupling that can potentially explain the measured extraordinary Tc.
The calculated Tc however does not match the experimental value when the phonons, which drive the superconducting mechanism here, are calculated in the standard harmonic approximation. It is considerably overestimated. In the harmonic approximation it is assumed that the force felt by the atoms is linear with the displacement from the equilibrium position, as in the well-known Hooke’s law. The harmonic approximation is generally valid if the atoms vibrate very close from the equilibrium position. Although high compression of H3S should limit the range of atomic displacement, the hydrogen atoms are significantly displaced from their equilibrium positions due to their very light mass, yielding to important effects beyond the harmonic approximation. Including the so-called anharmonic effects in the calculations, strong corrections were predicted for the phonons involving hydrogen atoms. Combining the anharmonic phonons with the electron-phonon interaction, the experimental Tc was reproduced. Thus, the researchers conclude that the record high-temperature superconductivity observed is due to the large interaction between electrons and anharmonic phonons.
The experimental result by Drozdov et al. and the theoretical explanation that followed [8] clearly address that very large superconducting Tc’s can be obtained even with the conventional electron-phonon pairing mechanism. Considering that this measurement is the first ever to have observed high Tc in hydrides, it is expected that many new experiments will follow. Will room-temperature superconductivity be observed under high-pressure in a different hydrogen-rich compound soon? Time will tell.
References
- A. P. Drozdov, M. I. Eremetes, and I. A. Troyan, “Conventional superconductivity at 190 K at high pressures”, arXiv:1412:0460. ↩
- M. Sakashita, H. Yamawaki, H. Fujihisa, and K. Aoki, “Pressure-Induced Molecular Dissociation and Metallization in Hydrogen-Bonded H2S Solid”, Phys. Rev. Lett. 79, 1082 (1997). ↩
- N. W. Ashcroft, “Metallic Hydrogen: A High-Temperature Superconductor?”, Phys. Rev. Lett. 21, 1748 (1968). ↩
- N. W. Ashcroft, “Hydrogen Dominant Metallic Alloys: High Temperature Superconductors?”, Phys. Rev. Lett. 92, 187002 (2004). ↩
- N. Bernstein, C. Stephen Hellberg, M. D. Johannes, I. I. Mazin, and M. J. Mehl, “What superconducts in sulfur hydrides under pressure and why”, Phys. Rev. B 91, 060511(R) (2015). ↩
- Defang Duan, Yunxian Liu, Fubo Tian, Da Li, Xiaoli Huang, Zhonglong Zhao, Hongyu Yu, Bingbing Liu, Wenjing Tian, and Tian Cui, “Pressure-induced metallization of dense (H2S)2H2 with high-Tc superconductivity”, Sci. Rep. 4, 6968 (2014). ↩
- J.E. Hirsch and F. Marsiglio, “Hole superconductivity in H2S and other sulfides under high pressure”, Physica C 511, 45 (2015). ↩
- Errea I., Chris J. Pickard, Joseph Nelson, Richard J. Needs, Yinwei Li, Hanyu Liu, Yunwei Zhang, Yanming Ma & Francesco Mauri (2015). High-Pressure Hydrogen Sulfide from First Principles: A Strongly Anharmonic Phonon-Mediated Superconductor, Physical Review Letters, 114 (15) DOI: http://dx.doi.org/10.1103/physrevlett.114.157004 ↩
3 comments
[…] [Continue reading at MappingIgnorance] […]
RBL would like to note that I have previously presented a continuum spinrevorbital motion that proposes a gravitomagnetic bonding between nuclei and superconducting electrons that may be used in the H2S to explain its high temperature superconductivity to predict novel binding hydrogen atoms and superconducting pairs during scattering of the superconducting electrons into continuum states (anharmonic states of vibrations) at the high temperatures and huge pressures within the lattice of H2S. . The scattering of superconducting e pairs was previously predicted to occur into such proposed continuum by disordered temperatures gradients and how such awkward phonons in p block elements would raise superconding temperature relative to the low superconducting temperatures in d block substances. I actually specifically predicted looking for such in lighter p block substances like sulfur and carbon! Such anharmonic or continuum scattering in H2S are explained by a gravitomagnetic chemistry involving hydrogen and sulfur atoms and novel gravitomagnetic binding of hydrogen and sulfur atoms into solids of greater densities at high pressures for magneto-gravimetric binding the scatter cooper pairs: as explained on page 14 and 20 {of “A Theory of the Relativistic Fermionic Spinrevorbital”} [http://www.academicjournals.org/journal/IJPS/article-abstract/592BAB049585 ] and even throughout this long article. But here on page 15 I explicitly note the ability of p block to invert phonons about continuum (anharmonic modes) relative to less phonon inversion about discontinuum (harmonic modes)! But such gravitomagnetic binding and spinrevorbitals thereof I proposed for transforming thermal, mechanical pressure, macro electric, gravity and macro magnetic energies and they bind fermions for explaining high temperature superconductivity and superfluidity as I occurs within H2S at high pressure! By my mechanism the high pressures on H2S are transformed to electric, gravitational and magnetic binding of the cooper pairs so they withstand higher temperatures. I proposed that such spinrevorbital states can bind and glue electrons and nuclei of elements like H and S at higher temperatures as in H2S and the surrounding electrons use the thermal energy, pressures and gravity within H2S to form the unparticles or spinrevorbital continuum of mass energy, momenta for glueing their superconducting fermions at the higher prevailing temperatures. The Copper pairs in H2S may be the condensation of gravitomagnetically bound electrons in H and S structures which with greater mass and magnetism rotate and fall in a spinrevorbitals of the continuum for for a superfluid motion of the Copper pairs which explains the need for higher pressures. But I applied and document such models to the mechanism of superconductivity and in so noted that heat and relative moving thermal energy relative to fermion can be transformed to gravito-magnetism (possibly at higher pressures) for nondissipation of motion of fermions for superconductivity and superfluidity! See the short article at this website : http://arxiv.org/abs/1412.4012 .
RBL (in a second longer article “A Theory of the Relativisitic Fermionic Spinrevorbital”) also considered in more details (a lot of details) how heat and relative thermal motions and pressure are transformed to gravity and gravity to magnetism by magnetic processes for forming continuum spinrevorbitals (unparticles) in p block materials like H2S and such would apply to hot pressurized objects like H2S for novel gravitomagnetic binding and chemistry at the high pressures and higher temperatures in analog to high pressure chemistry of diamond formation. I show in this article how such heat to pressure and pressure to electric and electric to gravity and gravity to magnetism is further intrinsically coupled to fermions for binding fermions at higher temperatures for novel magnetogravimetric molecules and superfluidity and superconductivity at higher critical temperatures. By this I first proposed in 2005 via sci-print archive and in 2012 via vixra archive and now in Jan 2015 via IJPS that quarks are held by a glue that is magnetic revolutionary (unparticle) for a gravitomagnetic basis of chemical bonding for holding the superfluidic hydrogen and Sulfur in nuclei together. In analog, I realized such weaker magnetic spinrevorbital hold electrons in atoms and molecules for superconductivity and superfluidity in orbitals and in continuum (forbidden states). I thereby realized that such continuum (anharmonic) spinrevorbitals in many atoms making up macro-objects can form by macroscopic heat, pressure, macro-electric, gravity, macromagnetic energies to form continuum states of varying mass, momenta and energies for binding macroobjects that constitute these anharmonic modes in H2S with high pressure induced superconductivity at higher temperatures. This is very beautiful!!! See a long article that ties everything together at this website : http://www.academicjournals.org/journal/IJPS/article-abstract/592BAB049585 . You can see the abstract at this website and you can also access the whole publication free of charge by clicking on ‘full text pdf’.
[…] Errea (2015) Unveiling the origin of the record superconductivityMapping […]