Flat heads
Flat heads

Phragmosis is a method by which an animal defends itself in its burrow by using its own body as a barrier. This term was coined by W.M. Wheeler in 1927 1 to describe a cryptic defensive technique employed by arthropods that use specially modified body structures to block nest entrances. A well example is the mygalomorph spider Idiosoma nigrum which protects itself in its tunnel blocking the entrance with its abdomen, which is unusually hard.
Phragmosis can also be observed in ants and they employ truncated body parts, usually the head but sometimes the abdomen. Phragmotic-headed ants, for instance, prevent intruders from entering nests by blocking the entrances or by pushing them out of entrance galleries. This defense mechanism has been described in several ants geni: Camponotus (subgenus Colobopsis and Hypercolobopsis), Colobostruma, Crematogaster (= Colobocrema), Pheidole, Oligomyrmex, Cephalotes and, recently, in Carebara2.

Phragmosis is particularly strongly developed in the New World arboreal genus Cephalotes, where usually all castes (queens, and major and minor workers) have highly adapted shield-like head morphologies that enable them to plug their nest entrance without exposing eyes, antennae or mandibles to any would-be intruders. Using this system, Soldiers act as living doors, closing off nest entrances with their heads when necessary. As an adaptation, they have a completely rimmed, saucer-shaped, cephalic disc on the heads. While the major workers’ most important tasks are chopping and transportation of larger prey and the defense of foraging trails and the nest, the main function of phragmotic workers is blocking tunnel entrances deterring intrusion of other predatory ants and invertebrates, acting therefore as a subcaste. The flattened heads of phragmotic major workers are a perfect fit for the tunnels created by wood-boring beetles in trees where these ants make their nest.
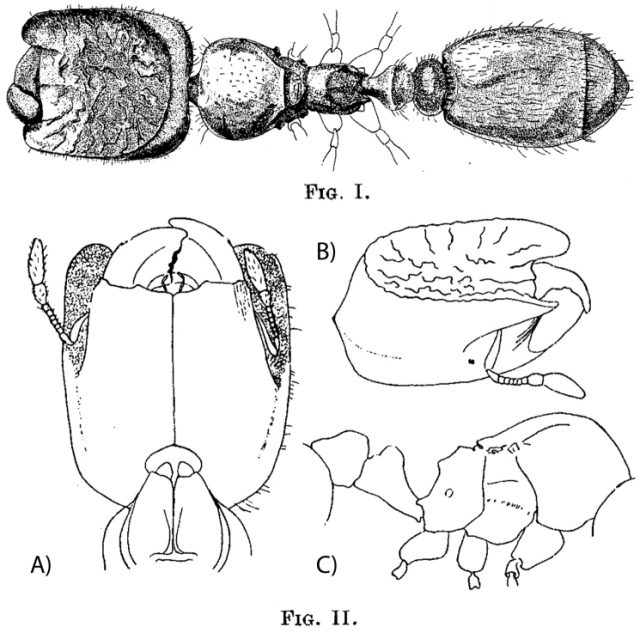
Often, the ants’ heads are camouflaged, either by a secretion of tangled fibers or by forest debris caught by small hairs. The secretion is formed by a fibrous material produced from thousands of pores that resemble glandular openings. This material coats and eventually accumulates in a dense tangle of material over the head disk. It is superficially similar to a mass of fungal filaments (mycelia) but does not have the branching pattern or internal structure indicative of a fungal origin. In other ant genera, phragmotic structures develop through the accretion of environmental debris held by specialized hairs that act like Velcro. The appearance of this material made the heads resemble the bark of a plant or the sand of the terrain and implied that the debris camouflaged this living gate to the nest. Since the encrusting material is similar to the foreign matter surrounding the area, it is difficult to distinguish it from the nest entrance and would effectively conceal it. If an unwanted visitor is detected, soldier ants lock themselves into a defensive position, blocking the tunnel entrances and making their nests harder to spot.
The used of accumulated debris as camouflage has been found in different species of insects, including basicerotine and stegomyrmicine ants. These are unusually slow, dirty ants that freeze when disturbed. Thus, camouflage is enhanced by a lack of motion, also in the case of the phragmotic ants protecting the entrances since guards remain stationary there. 3
Reversed phragmosis was described for the queen of Pheidole embolopyx, which has a peculiar posteriorly truncated gaster with a rear surface covered with unusual hook-shaped hairs. Furthermore, the queen’s antennal scapes, anterior clypeal border, and frontal carinae are covered by gelatinous sheaths, varying in size and shape among individuals and on the same queen over time, and often absent altogether. Contrary to expectation, P. embolopyx queens do not use the gaster to block nest entrances or otherwise to repel intruders directly, nor do the gelatinous structures seem to be especially attractive or repellent to the workers.
There is also intranidal phragmosis in ants4. The ergatoid queen of Blepharidatta conops (Formicidae, Myrmicinae) blocks the entrance of the brood chamber. Mature nests of these ants are short blind vertical cylinders, generally excavated directly in the ground. Nurse workers hold the brood in the mandibles while resting along the nest’s walls. When nests are visited or inhabited by myrmecophiles and/or predators, ant workers hide the larvae in the nest’s subsidiary chamber, the entrance of which is then blocked by the peculiar phragmotic disk of the single queen in the colony.
Phragmosis is a non-aggressive, non-violent way of protecting the nest.
References
- Wheeler WM (1927) The physiognomy of insects. Q Rev Biol 2: 1–36. ↩
- Fischer G, Azorsa F, Garcia FH, Mikheyev AS, Economo EP (2015) Two new phragmotic ant species from Africa: morphology and next-generation sequencing solve a caste association problem in the genus Carebara Westwood. Zookeys (525): 77-105. DOI: 10.3897/zookeys.525.6057 ↩
- Wheeler DA, Hölldobler B (1985) Cryptic phragmosis: the structural modifications. Psyche 92(4): 337-353. ↩
- Brandão CRF, Diniz, JLM, Silva PR, Albuquerque NL, Silvestre R (2001) The first case of intranidal phragmosis in ants. The ergatoid queen of Blepharidatta conops (Formicidae, Myrmicinae) blocks the entrance of the brood chamber. Insectes Soc. 48: 251-258. ↩
2 comments
[…] Posted in Biology, Noticias, Science | 0 comments […]
Excellent Article, Excellent Blog , Excellent Site ✅✅✅