The enigmatic tetrahalodiboranes
The enigmatic tetrahalodiboranes
Among the group 13 elements, boron alone forms halides of the type X2B-BX2 (X=F, Cl, Br, I), known as tetrahalodiboranes. Over the past five years, the filing of patents involving tetrahalodiboranes has outpaced their appearance in journal articles at a rate of two to one, mostly due to the current interest in B2F4 for the doping of silicon with B+ ions in order to be used in the fabrication of semiconductor devices.
Tetrahalodiboranes are enigmatic compounds with a rich history, but their synthetic chemistry is little explored. Unlike for their synthetically useful tetrasubstituted counterparts, synthetic interest in tetrahalodiboranes ebbed after an initial spike in their use in the mid-20th century. Given their liability and outstanding reactivity potential, the relative absence of literature on tetrahalodiboranes is almost certainly due to their historically difficult preparation, which in many cases involves gas-phase synthetic steps.
Very recently, however, a team from Julius-Maximilians-Universität Würzburg (Germany) presented 1a simple solution-phase syntheses of B2X4 (X=F, Cl, I) from B2Br4, rendering all four tetrahalodiboranes considerably more accessible in practical amounts (Figure 1, top).
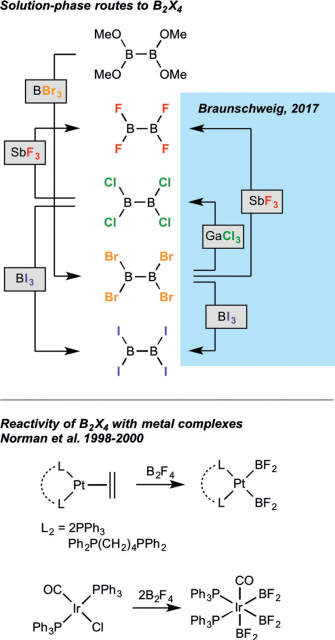
Even though the coordination chemistry of diboron units with transition metals is rather well-developed and diverse, the only known attempts to combine tetrahalodiboranes by Norman et al 23 with transition-metal fragments were made with B2F4 (Figure 1, bottom).
Now, a team that includes researchers from Würzburg and Ikerbasque researcher at DIPC Eduard Matito, investigate 4 what they thought was a more interesting reactivity for electron-rich transition-metal fragments with the heavier tetrahalodiboranes. Their efforts led to the isolation of a highly unusual diplatinum complex containing two boryl ligands and a bridging [B2I4] ligand best described as a diboranyl dianion.
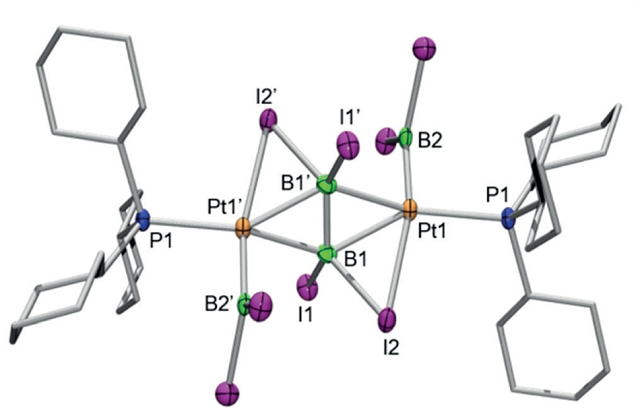
The experimental work and theoretical calculations revealed a highly unusual structure based on two [Pt(BI2)(PCy3)] fragments – where PCy3 is tricyclohexylphosphine, a tertiary phosphine with the formula P(C6H11)3 – bridged via a single B2I4 unit through a network of interactions of the Pt centers with B-I and B-B σ bonds (figure 2). Remarkably, the B2I4 unit can be seen as an olefin analogue.
This compound is the first transition-metal complex of an intact B2X4 (X=halide) species of any type, and the first example of transition metal coordination to a reduced diborane species. These results provide a glimpse of the potentially exciting coordination chemistry of tetrahalodiboranes, about which very little is currently known.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance.
References
- M. Arrowsmith, J. Böhnke, H. Braunschweig, A. Deißenberger, R. D. Dewhurst, W. C. Ewing, C. Hçrl, J. Mies, J. H. Muessig, Chem. Commun. 2017, 53, 8265–8267. ↩
- A. Kerr, T. B. Marder, N. C. Norman, A. G. Orpen, M. J. Quayle, C. R. Rice, P. L. Timms, G. R. Whittell, Chem. Commun. 1998, 319–320. ↩
- N. Lu, N. C. Norman, A. G. Orpen, M. J. Quayle, P. L. Timms, G. R. Whittell, Dalton Trans. 2000, 4032–4037 ↩
- Holger Braunschweig, Rian D. Dewhurst, J. Oscar C. Jiménez-Halla, Eduard Matito, and Jonas H. Muessig (2018)Transition-Metal p-Ligation of a Tetrahalodiborane Angew. Chem. Int. Ed. doi: 10.1002/anie.201709515 ↩
1 comment
[…] Artikulu akademiko baino patente gehiago erregistratzen dira tetrahalodiboranoen inguruan, duten interes teknologiakoa dela eta. Haien kimika, baina, misterio bat da. DIPCko jendearen The enigmatic tetrahalodiboranes […]