Metal substrates in catalytic reactions
Metal substrates in catalytic reactions
When we consider the concepts metal and catalysis, we tend to assume quite matter-of-factly that the metal will be the catalyst. This assumption is based on the fact that metals can be found in reactions where they act as catalysts or co-catalysts in the form of coordination and organometallic compounds, nano-sized or bulk materials and metalloenzymes. Obviously, this does not mean that metals can not be substrates: biological evolution has devised numerous enzymes capable of catalytically transforming metals. Still, metal substrates are very rarely employed in catalytic reactions. This is about to change.
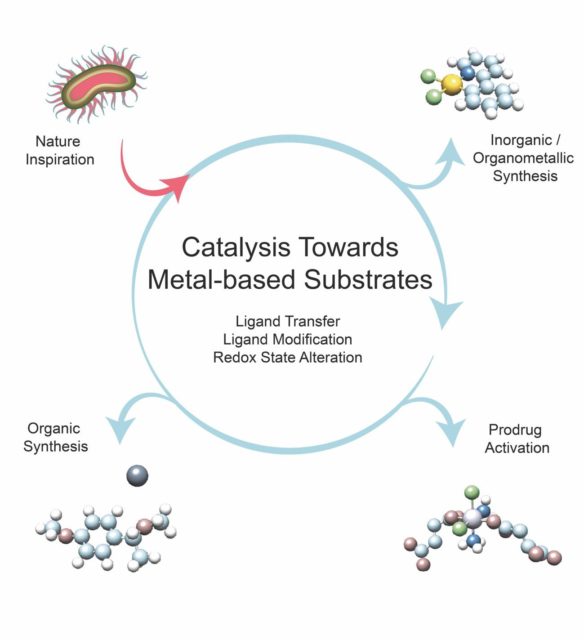
In this context, we need to understand metals not as pure substances but as compounds. Hence, a more appropriate term is metal substrates. Metal substrates are chemical substances such as metal salts (e.g. halides) or oxides and coordination or organometallic complexes, whose coordination sphere and/or redox state is altered by a catalyst in multiple turnovers.
As organic synthesis shows, control of reaction kinetics through catalysis can translate into unique reactivity and selectivity, affording otherwise inaccessible products. Likewise, designing catalysis schemes for the transformation of metal substrates could result in efficient and sustainable processes for novel inorganic compounds and materials. Besides, catalytic control of the redox chemistry of metals may help tune their reactivity in different chemical environments, thus fostering new applications. Consequently, the conversion of metal substrates to added-value compounds may mean a gigantic step forward in catalysis, opening up new opportunities for technology applications of inorganic chemistry.
Now, Fernando López Gallego and Luca Salassa have teamed up to describe 1 the novel synthetic paths and reactivities opened by catalytic transformations of metal substrates. In the process, they identify the opportunities and challenges for the implementation of this approach to chemical manufacturing or biomedicine, among other potential applications.
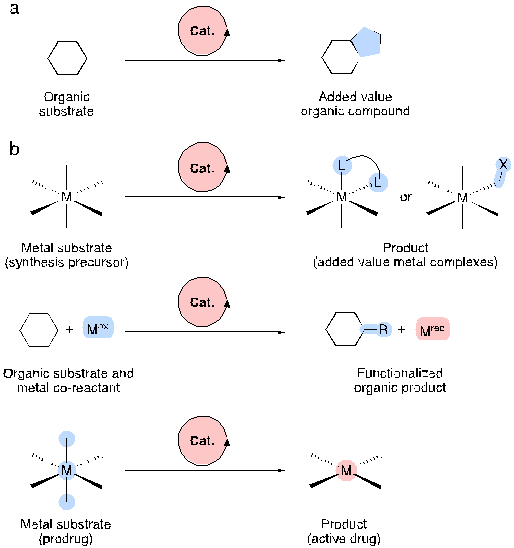
The authors focus on a selection of catalytic transformations that make use of metal-containing species as substrates, chosen because of their significance in synthetic inorganic and organic chemistry, biology and medicine. They start, however, with a brief description of how several natural organisms successfully manage the catalytic transformation of metals for their own benefit. Overall, the examples highlighted unveil how the conversion of metal substrates to added-value compounds can result in a paradigm shift in catalysis.
In this shift, biomimetics is key. Since the capacity of living systems to bioassimilate metals via redox reactions is outstanding, it should be taken as an inexhaustible source of inspiration for developing novel catalysis schemes capable of transforming a variety of metal forms (solids, complexes, soluble salts) and expanding the (bio)inorganic toolbox for diverse technological applications (i.e. chemical manufacturing, biomedicine, metal mining/recovery). This exciting challenge requires the merging of knowledge and expertise from diverse fields, including inorganic/organometallic chemistry, environmental science, biochemistry, and microbiology.
More on the subject:
Bioorthogonal catalytic activation of anticancer metal complexes
A hydrogel matrix as viable solution for the efficient catalytic activation and delivery of cisplatin
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may have been copied verbatim or almost verbatim from the referenced research papers.
References
- Fernando López Gallego and Luca Salassa (2022) Catalysis Towards Metal-Based Substrates: A New Prospect For Inorganic Chemistry Chem Catalysis doi: https://doi.org/10.1016/j.checat.2022.10.0 ↩