The need for experiments in working conditions: the case of carbon monoxide oxidation
The need for experiments in working conditions: the case of carbon monoxide oxidation
Carbon monoxide (CO) oxidation (CO + ½ O2 → CO2) is an industrially relevant catalytic reaction, and accordingly, it is among the most studied processes in fundamental surface science. This has led to a wealth of research studies in vacuum, but the doubts remain on whether this reaction proceeds in the same way when changing to realistic working conditions.
Actually, the majority of scientific investigations have been performed with either single crystals or under ultra-high vacuum, far from the requirements of industry, this is the “pressure gap”. Therefore, an extrapolation of their conclusions to real catalytic systems (atmospheric pressures and powder/nanoparticle catalysts) may not be correct.
There is also a “structure gap” due, first, to the fact that metallic nanoparticles comprise several facets, and monitoring their specific activity and interplay during a chemical reaction is challenging; and, second, because the information that can be obtained using conventional flat crystals is restricted to one plane alone, and hence it is not representative of the structure of a real catalyst.
With the advent of powerful analytical techniques that operate at high gas pressures, such as Ambient Pressure X-ray Photoemission (AP-XPS), the “pressure gap” has been reduced significantly. However, a “structure gap” persists, since most AP-XPS studies are carried out using single crystal surfaces, which contrast with the multi-facet structure of technologically relevant nanoparticles.
One approach to partially close this structure gap is to use cylindrical sections of single crystals, since their curved surface provides a smooth variation of the crystal orientation that allows to systematically compare different facets under the very same reaction conditions (Figure 1).
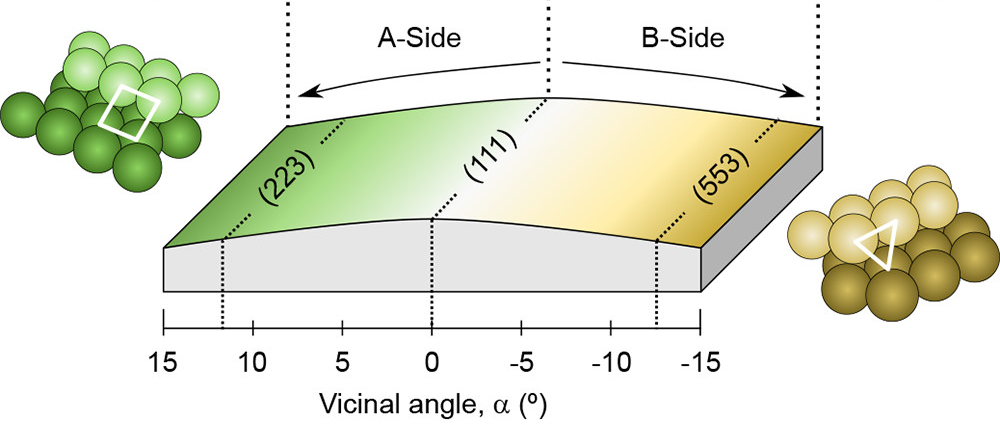
Figure 1 shows a Rh crystal “curved” around the (111) direction, leading to a smooth variation of the crystal plane at each point. Such geometry allows the different crystal facets to be sequentially probed by scanning the X-ray beam in AP-XPS. With this approach, researchers have investigated 1 the effect of A-type (square geometry, left side of the crystal) and B-type (triangular geometry, right side of the crystal) atomic packing of steps on the catalytic CO oxidation on Rh at millibar pressures.
In this work, the team has exposed the Rh curved sample of Figure 1 to a reactive CO+O2 atmosphere, and has identified the sequence and thermal evolution of surface chemical species across the different crystal planes around the reaction light-off. A striking asymmetry is observed between A- and B-type steps, both during the ignition and at the fully active reaction stages (Figure 2).
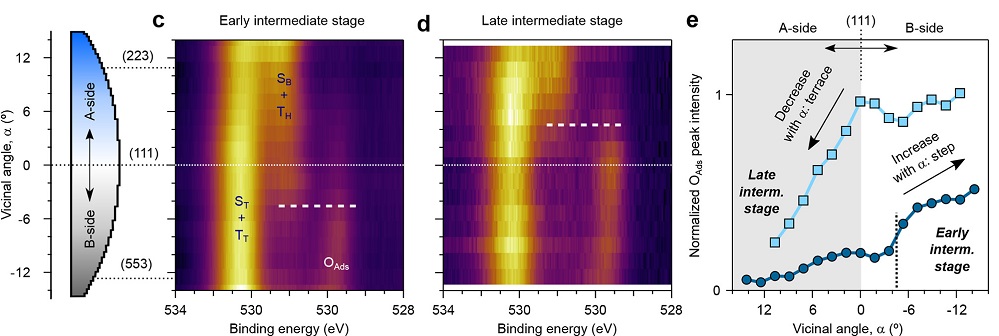
Figure 2 shows XPS maps of chemical species at different temperatures as a function of the local crystal plane. The maps acquired during the ignition prove the partial CO-depletion and O-accumulation at B-steps, triggering the earlier B-step ignition. In the active stage of the reaction, the low-active Rh oxide builds up at A-steps (square atomic packing), while B-steps (triangular atomic packing) retain the active chemisorbed oxygen species during reaction conditions.
Therefore, the researchers conclude, B-steps are more active toward the CO oxidation than A-steps. These experiments using curved surfaces emphasize the need for studies in working conditions on the influence of steps and their interplay with other surface sites in chemical reactions.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may be copied verbatim or almost verbatim from the referenced research papers.
More on the subject:
Catalysis depends on the crystallographic plane of the catalyst
Simultaneous ignition of the CO oxidation on a curved platinum surface
References
- Fernando García-Martínez,Lisa Rämisch,Khadiza Ali,Iradwikanari Waluyo,Rodrigo Castrillo Bodero, Sebastian Pfaff, Ignacio J. Villar-García, Andrew Leigh Walter, Adrian Hunt, Virginia Pérez-Dieste, Johan Zetterberg, Edvin Lundgren, Frederik Schiller, and J. Enrique Ortega (2022) Structure Matters: Asymmetric CO Oxidation at Rh Steps with Different Atomic Packing J. Am. Chem. Soc. doi: 10.1021/jacs.2c06733 ↩