Alzheimer’s disease: Is amyloid beta 42 the answer?
Alzheimer’s disease: Is amyloid beta 42 the answer?
Authors: Andrea Sturchio, MD, PhD Student, Clinical Neuroscience, Karolinska Institutet; Kariem Ezzat, Research Scientist, Laboratory Medicine, Karolinska Institutet, and Samir EL Andaloussi, Professor, Laboratory Medicine, Karolinska Institutet
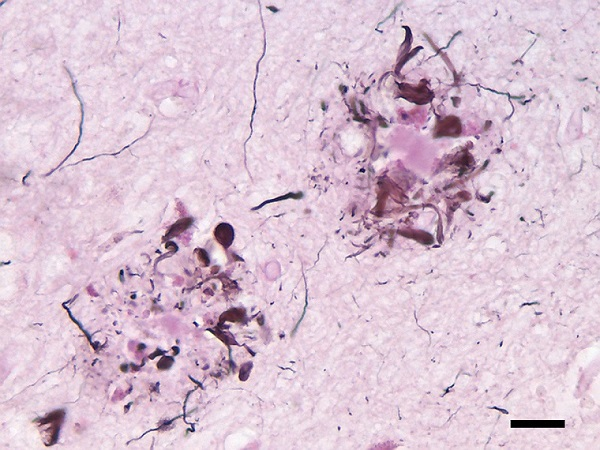
In 1906, Alois Alzheimer, a psychiatrist and neuroanatomist, reported “a peculiar severe disease process of the cerebral cortex” to a gathering of psychiatrists in Tübingen, Germany. The case was a 50-year-old woman who suffered from memory loss, delusions, hallucinations, aggression and confusion – all of which worsened until her untimely death five years later.
In the autopsy, Alzheimer noticed distinctive plaques on her brain. These plaques – clumps of amyloid-beta protein – are still considered to be the cause of Alzheimer’s disease.
However, this theory has two major problems. First, it does not explain why many subjects (even old people) have plaques in their brains in the absence of any neurological symptoms, such as memory loss. Second, clinical trials for drugs that reduce these plaques have been unsuccessful – with one recent exception, but more of that later.
When amyloid-beta protein accumulates in the form of plaques (insoluble clumps), the original soluble form of the protein, which performs important functions in the brain, is consumed and lost. Some studies have shown that reduced levels of soluble amyloid-beta – called amyloid-beta 42 – have led to patients having worse clinical outcomes.
The cause of Alzheimer’s disease
In a recent study, published in the Journal of Alzheimer’s Disease, we investigated whether it’s the amount of plaques in the brain or the amount of amyloid-beta 42 remaining that is more important for Alzheimer’s disease progression.
To answer this question, we studied data on a group of people who have a rare inherited gene mutation that puts them at high risk of developing Alzheimer’s disease. The participants were from the Dominantly Inherited Alzheimer Network cohort study.
We found that the depletion of amyloid-beta 42 (the functional version of amyloid-beta) is more harmful than the amount of plaques (the insoluble clumps of amyloid beta).
Participants had an average of three years follow-up and we found that those with high levels of amyloid-beta 42 in their cerebrospinal fluid (the liquid around the brain and spinal cord) were protected and their cognition was preserved over the study period. This chimes with many studies that showed important functions of amyloid-beta 42 in memory and cognition.
It is also relevant because we studied people with the genetic mutation who develop Alzheimer’s disease, a group that is considered to provide the strongest evidence supporting the idea that amyloid-beta plaques are harmful. However, even in this group, those with higher cerebrospinal fluid (CSF) levels of amyloid-beta 42 remained cognitively normal regardless of the amount of plaques in their brains.
It is also worth mentioning that in some rare, inherited forms of Alzheimer’s disease – for example, in carriers of the so-called Osaka gene mutation or Arctic mutation – people can develop dementia having low levels of amyloid-beta 42 and no detectable plaques. This suggests that plaques aren’t the cause of their dementia, but low levels of amyloid-beta 42 might be.
Lecanemab – the one recent exception
How will our findings affect drug development and clinical trials for Alzheimer’s disease? Until the recent trial with lecanemab, an antibody drug that reduces plaques, all the drug trials in Alzheimer’s disease have failed.
Some drugs were designed to reduce the levels of amyloid-beta 42, based on the rationale that if levels of the normal protein are reduced, patients will accumulate fewer plaques. Unfortunately, these drugs often made the patient’s condition worse.
Lecanemab was recently reported to have a small but significant effect in reducing cognitive decline. According to previous studies, this drug increases the levels of amyloid-beta 42 in the CSF. This is, again, in line with our hypothesis, namely that the increase of the normal amyloid protein can be beneficial.
We will know more when the results of the lecanemab trial are published. At the moment, all we have is a press release from the makers of the drug.
We think that it will be important for future trials to focus on the levels of amyloid-beta 42, and whether it is beneficial to increase and restore its levels to normal values instead of targeting it for removal. This could be achieved using proteins similar to amyloid-beta 42 – so-called “protein analogues” – but that clump together less than the natural ones.
This active protein replacement approach might become a promising new avenue of treatment for Alzheimer’s and other protein aggregation diseases, such as Parkinson’s and motor neuron disease.![]()
More on the subject:
Single brain scan can diagnose Alzheimer’s disease
Alzheimer’s disease: type 3 diabetes?
This article is republished from The Conversation under a Creative Commons license. Original article.
1 comment
These findings are compatible with the Amyloid Beta Dysfunction Hypothesis (H. Hillen, Frontiers in Neuroscience, 2019). According to this hypothesis early misfolding creates physiological Amyloid beta monomer deficiencies at synaptic sites. In a kind of endogenous repair mechanism, the synapse starts compensation of Abeta monomer production by upregulation of BACE. A useful therapeutic approach in prevention and early treatment of Alzheimer’s Disease should focus on removal of early soluble misfolded Amyloid beta species to break and reverse vicious circle of enhanced Amyloid beta production.