A formidable characterization tool for one-dimensional metal−organic chains
A formidable characterization tool for one-dimensional metal−organic chains
One-dimensional metal−organic chains often possess a complex magnetic structure, susceptible to modification by alteration of their chemical composition. The possibility to tune their magnetic properties provides an interesting playground to explore quasi-particle interactions in low-dimensional systems. These systems have potential applications in the fabrication of nanodevices for spin sensing, spintronics, quantum computing based on the spin degree of freedom, and high-density information storage. Magnetism in low-dimensional systems is also interesting from a fundamental point of view.
One-dimensional magnetic metal−organic coordination chains are types of molecular nanomagnets that can be considered as candidates for building single-chain magnets under certain conditions. Currently, a complete understanding of the relation between the chemical composition of metal−organic systems (defined by the type of organic ligand and magnetic atom constituents) and relevant magnetic parameters is still missing.
However, the study of low-dimensional magnetic systems, often exhibiting strong electron correlations, is far from trivial due to the limits of available experimental and theoretical tools. Not that the experimental techniques that could be used are not powerful ones, but the accurate determination of magnetic properties from the acquired spectra is possible only with the help of complex theoretical many-body calculations.
The use of molecules of a bright green paramagnetic solid called nickelocene may change this situation. Nickelocene is a metallocene. Metallocenes usually adopt structures in which a metal ion is sandwiched between two parallel cyclopentadienyl (Cp) rings. Nickelocene is the popular name of bis(cyclopentadienyl)nickel, Ni(C5H5)2, NiCp2 for short.
A very interesting option to study low-dimensional magnetic systems is based on the use of inelastic electron tunneling spectroscopy (IETS), which permits a local probe of both vibrational and spin excitations. As it is used now, the technique has clear limitations, as the strong interaction with itinerant electrons in the metallic electrodes makes the inelastic tunneling channels of most magnetic adsorbates extremely weak. This is what nickelocene resolves.
Using NiCp2-decorated tips would allow detecting magnetic exchange interactions with spins of individual atoms at surfaces via the spin excitations of the NiCp2 probe and their mutual coupling. But, does it work? And, how does it compare with other experimental-theoretical combos?
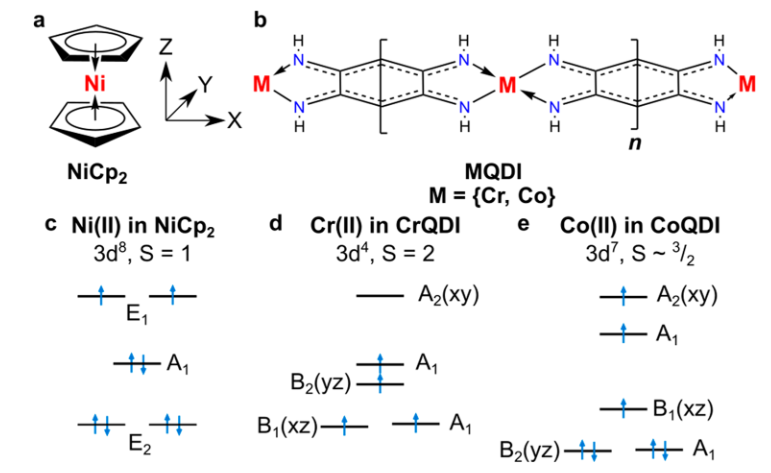
Now, a team of researchers provides 1 a thorough analysis of the magnetic structure of long, isostructural metal−organic 1D coordination polymer chains grown on a gold surface, Au(111), by using multiple experimental and theoretical techniques. The chains of choice were the product of the reaction of transition metal (Cr, Co) atoms with 2,5-diamino-1,4-benzoquinonediimin.
By a combination of these experimental and theoretical methods, the team can unravel the magnetic properties of the metal−organic chains. Specifically, the researchers can determine the spin multiplicity and the magnetic anisotropy of the chains.
They can also give a reason as to why NiCp2-IETS does not exhibit exchange splitting despite probing a magnetic center. Because they found that IETS spectroscopy acquired with a NiCp2-decorated tip is a formidable characterization tool. The scientist concude that this technique, combined with an appropriate theoretical analysis, can provide detailed information about the local magnetic properties.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may have been copied verbatim or almost verbatim from the referenced research papers
References
- Christian Wäckerlin, Aleš Cahlík, Joseba Goikoetxea, Oleksandr Stetsovych, Daria Medvedeva, Jesús Redondo, Martin Švec, Bernard Delley, Martin Ondráček, Andres Pinar, Maria Blanco-Rey, Jindřich Kolorenč, Andrés Arnau, and Pavel Jelínek (2022) Role of the Magnetic Anisotropy in Atomic-Spin Sensing of 1D Molecular Chains ACS Nano doi: 10.1021/acsnano.2c05609 ↩