Triple-decker antiaromatic π-stacking leads to a liquid crystal
In organic chemistry, π-stacking systems are supramolecular structures that arise due to the dispersion force, a type of intermolecular noncovalent interaction. They are a common occurrence in nature. The stabilized structure of DNA is a very prominent example of a π-stacking system, and so are the arrangement of amino acids in certain proteins.
Interestingly, π-stacking can be leveraged in the design of materials with useful electronic and optical properties. These include organic semiconductors of various kinds, as well as conjugated polymers for sensing and biomedical applications.
Thus far, a good portion of technologically relevant π-stacking systems have been limited to aromatic compounds, which have inherent π-electron clouds. On the other hand, antiaromatic compounds, though promising candidates for developing electric conductors, have rarely been reported as the building units of π-stacking systems.
Surprisingly, in a recent study, a research team led by Professor Hiromitsu Maeda from Ritsumeikan University, Japan, reported a novel antiaromatic π-stacking system that enabled the formation of a highly conductive liquid crystal 1.
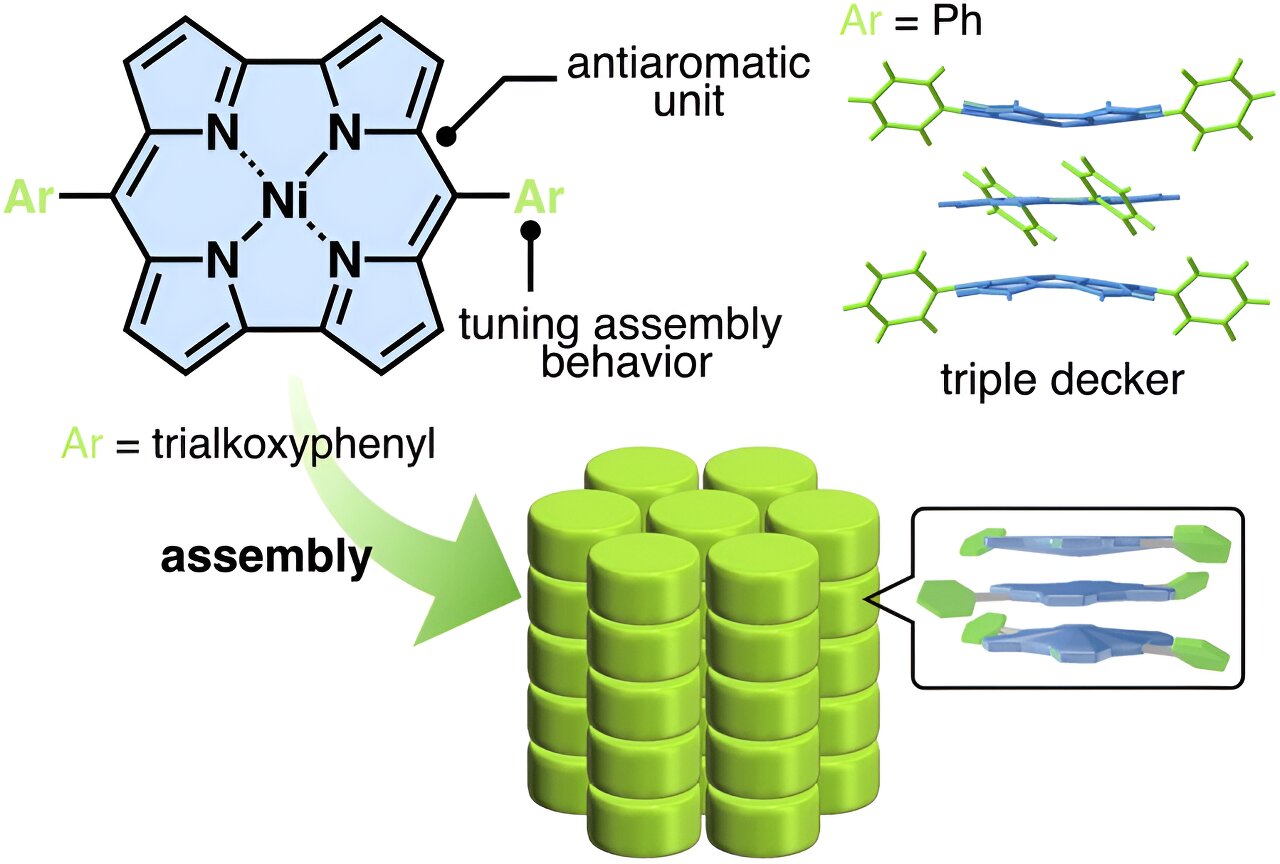
The reported compounds in question are NiII-coordinated norcorroles with modified aryl moieties as side chains. Previously, achieving π-stacking in similar norcorroles failed because hydrogen-bonding interactions between the side chains opposed the face-to-face stacking of the planar antiaromatic units. This time, however, the research team had an ingenious idea.
“We hypothesized that the introduction of side interacting moieties with less directionality would enhance the stacking between norcorrole units,” explains Prof. Maeda. “Thus, we attempted the simple introduction of aliphatic chains, which induce van der Waals interactions. These interactions can be effective for modulating the stacking structure of a material.”
As evidenced through various experiments and molecular dynamics simulations, the proposed strategy worked as intended. The norcorrole units formed columnar structures through the stacking of arrangements known as “triple-decker.” In these arrangements, a planarized molecule is sandwiched between two slightly bowl-shaped molecules.
Using the proposed molecular design, the researchers then synthesized liquid crystals. Thanks to the triple-decker stacking, a liquid crystal exhibited remarkable electric conductivity as well as thermotropicity; that is, an order parameter that depends on temperature.
“The control of molecular interactions based on molecular design and synthesis, as demonstrated in our study, will be crucial for future applications,” says Prof. Maeda. “Properties such as high electric conductivity in liquid crystals may be used for the fabrication of electronic devices. In addition, stimuli-responsive behaviors in soft materials can be used to modulate relevant properties, like photoluminescence, according to pressure and temperature.”
Taken together, the findings of this study bring to light a promising strategy for designing new compounds based on molecular assemblies of antiaromatic units. With any luck, this will open up new avenues for materials design, ultimately leading to better organic electronics, optoelectronics, and sensing devices.
References
- Ishikawa, S. et al (2024) Norcorroles as antiaromatic π-electronic systems that form dimension-controlled assemblies Chemical Science doi: 10.1039/D4SC01633E ↩