Neural stem cells discovered outside the mouse brain
Neural stem cells discovered outside the mouse brain
Author: José R. Pineda earned his Ph.D. from the University of Barcelona in 2006. Since 2007, he has conducted research at Institut Curie and the French Alternative Energies and Atomic Energy Commission. He is currently a researcher at the University of the Basque Country – EHU, where his work focuses on the role of stem cells in both physiological and pathological contexts.
For decades, biology textbooks have repeated the same idea with almost ritual certainty: neural stem cells, the cells that fuel the birth of neurons and glia during development, live exclusively inside the brain and spinal cord. They are the architects of the central nervous system, and nowhere else. It has always seemed like an elegant rule of nature—clean, simple, and unbreakable. But science rarely stays still, and sometimes a single unexpected observation is enough to make a familiar landscape suddenly look foreign. A study 1 done by Hang and collaborators published in Nature Cell Biology has done exactly that. A large international team has uncovered a population of neural stem cells located far beyond the borders of the central nervous system. They are not neural crest progenitors, nor reprogrammed cells, nor artefacts of culture. They are bona fide neuroepithelial‑derived stem cells, essentially the same type that builds the embryonic brain, quietly residing in tissues such as the lung, tail, dorsal root ganglia, and embryonic limb of the mouse. And that discovery forces us to rethink some of the most basic assumptions about how and where the nervous system forms.
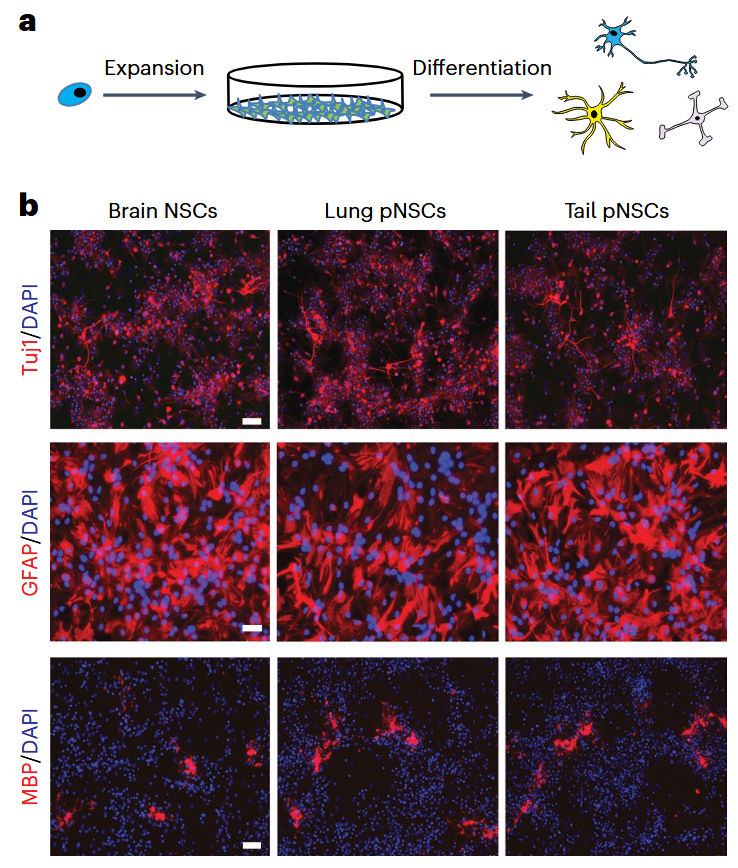
The story begins with a serendipitous twist. The researchers were attempting to replicate a set of controversial (and later retracted!!) experiments claiming that exposing cells to a low‑pH shock could convert them into pluripotent stem cells. Their goal was simply to verify those claims. Instead, what they found were small clusters of cells that looked remarkably like neural stem cells, even though the tissue of origin was not the brain. The result did not match the original hypothesis, but it pointed to something far more intriguing. Perhaps these cells had not been created by the treatment; perhaps they had been there all along. That possibility changed the direction of the project. The team began systematically searching for these mysterious cells using transgenic mice in which neural stem cells glow under the microscope. Immediately, unexpected patterns began to appear. In the adult lung, for instance, a rare population of cells (peripheral neural stem cells abbreviated as “pNSCs”) lit up with the same fluorescent (SOX1-GFP+) markers that define neural stem cells in the hippocampus. When the researchers isolated them and grew them in culture, they behaved just like classical neural stem cells: they self‑renewed, formed stable colonies, and differentiated into neurons, astrocytes, and oligodendrocytes. Even more striking, when transplanted into the developing mouse brain, they integrated into the tissue, survived for weeks, and matured into functional neural cell types without forming tumors.
For decades, scientists have known that most neural‑like cells found outside the central nervous system come from the neural crest, a migratory population that gives rise to the peripheral nervous system. Yet these newly discovered cells didn’t fit that category. They lacked the molecular signatures of neural crest derivatives and, when the researchers used genetic tools to permanently mark all descendants of the neural crest, the new cells remained unmarked. That meant they had to come from a different origin. The breakthrough came with another lineage‑tracing experiment, this time using a mouse line that permanently labels neuroepithelial cells (these cells are the earliest progenitors of the central nervous system). The result was unambiguous: the peripheral stem cells were marked. They were descendants of neuroepithelial cells, just like the neural stem cells in the brain. The only difference was “geographical”. Somewhere early in development, a subset of these neuroepithelial progenitors seems to leave the neural tube and migrate into other tissues, where they settle, lie low, and remain as a hidden reservoir of neural stem cells.
To understand exactly who these cells are (and who they are not!) the researchers turned to single‑cell RNA sequencing, which allows thousands of individual cells to be profiled one by one. The comparison between brain‑derived neural stem cells and these peripheral stem cells was surprisingly tight. Their gene expression programs, transcriptional signatures, and developmental potentials overlapped almost perfectly. There were small differences, subtle enough to reflect the influence of their local environments rather than a distinct lineage identity, but the overall conclusion was crystal clear: these peripheral stem cells are genuine neural stem cells, not impostors and not a hybrid class. Moreover, the study found them in tissues like lung and tail, but not in others, such as the heart, where only fully differentiated neurons were detected. This selective presence suggests that their migration during development follows precise anatomical routes and that their persistence into postnatal life is controlled by finely tuned local niches. Many of these cells exist in a quiescent state (dormant, where cells only divide rarely) while others show signs of activation, hinting at an internal balance similar to what is seen in the neural stem cell compartments of the brain.
From the functional point of view these peripheral stem cells are far from decorative. The researchers showed that during embryonic and even postnatal development, these cells contribute to forming mature neurons in multiple tissues, including dorsal root ganglia and intestine. In some organs, their contribution is confined to early development, while in others it continues after birth. This suggests that the body maintains a much more distributed neurogenic potential than previously appreciated, opening the door to new concepts about how peripheral organs might regulate their own innervation. Thus, the implications of this discovery are broader and profound: If neural stem cells exist outside the central nervous system, why are they there? What purpose do they serve? Could they play a role in tissue repair after injury, or in adapting organ function to different physiological needs? Are they remnants of an ancient developmental program, or part of an underappreciated regulatory system? These questions remain open, but the answers could reshape our understanding of neural development and regeneration.
From a translational perspective, the idea is tantalizing, because access to neural stem cells within the brain is difficult and risky, however, peripheral tissues are far more accessible. If these newly identified stem cells can be harnessed, they could someday provide a source of neurons for repair therapies without the need for invasive brain surgery. This is, of course, speculative, and the present study does not claim any therapeutic application. But it establishes a conceptual foundation that future research will almost certainly build upon. What stands out most in the work of Han and collaborators is the elegance of its challenge to a long‑held belief. Scientific dogmas often persist not because they are unquestioned, but because no evidence has emerged to contradict them. Here, through careful experimentation and a willingness to follow unexpected results, the authors reveal that the nervous system’s developmental boundaries are more porous and more flexible than we thought. They uncover a kind of biological quiet zone: a dispersed, semi‑hidden network of neuroepithelial‑derived stem cells that sit outside the brain yet retain the full potential to act like neural stem cells.
This discovery does more than update the detail of developmental biology. It rewrites part of the map. It reminds us that the body is full of overlooked corners, of wanderers and settlers that defy the categories we impose on them. And it highlights a truth that recurs again and again in science: nature does not draw lines where we expect them. Sometimes, it leaves doors open, quietly, subtly, waiting for us to notice…
References
- Dong Han, Wan Xu, Hyun‑Woo Jeong, Hongryeol Park, Kathrin Weyer, Yaroslav Tsytsyura, Martin Stehling, Guangming Wu, Guocheng Lan, Kee‑Pyo Kim, Henrik Renner, Dong Wook Han, Yicong Chen, Daniela Gerovska, Marcos J Araúzo‑Bravo, Jürgen Klingauf, Jens Christian Schwamborn, Ralf H Adams, Pentao Liu, Hans R Schöler (2025) Multipotent neural stem cells originating from neuroepithelium exist outside the mouse central nervous system Nat Cell Biol. doi: 10.1038/s41556-025-01641-w. ↩