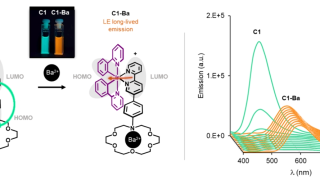
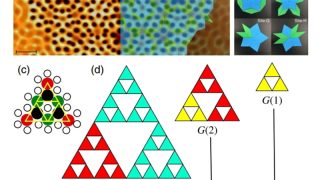
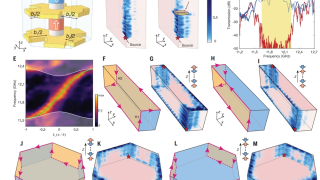
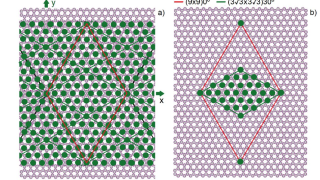
Zooming through graphene: The strange world of electron transit time
Imagine sending a tiny particle, like an electron, through a material so thin it’s just a single layer of atoms. You’d expect it to take a certain amount of time to pass through, right? Maybe a little longer if the material gets thicker, or shorter if it’s super thin. But what if the electron seemed […]