New hope against tuberculosis: spectinamides
New hope against tuberculosis: spectinamides
Tuberculosis (TB) represents a serious public health problem. The latest reports estimate an incidence of 8.7 million cases in 2001 and over 1.4 million deaths per year. The disease is usually be treated with a first-line drugs, isoniazid and rifampin, the two most potent anti-tuberculosis agents. However, the prevalence of multidrug-resistant (MDR) tuberculosis cause by Mycobacterium tuberculosis isolates that are resistant to both drugs is rising. In 2011, WHO estimates roughly 630.000 cases of MDR tuberculosis worldwide. After initial reports in 2006, extensively drug-resistant (XDR) tuberculosis (defined as tuberculosis cause by isolates resistant to isoniazid, rifampin, fluoroquinolones, and any of the second-line drugs such amikacin, kanamycin or capreomycin) has been confirmed in 84 countries, heralding the possibility of virtually untreatable tuberculosis. The emergence of MDR and XDR strains, some resistant to all available drugs, underscore the need for new anti-tubercular drug candidates.

With the participation of our Spanish colleague J. A. Ainsa (Department of Microbiology, University of Zaragoza), it has been recently published in Nature Medicine 1the description, synthesis and evaluation of a new class of semisynthetic spectinomycin analogs, the spectinamides, with excellent narrow-spectrum anti-tubercular activity. Spectinomycin is a classical aminoglycoside antibiotic produced by the bacterium Streptomyces spectabilis, it is a potent bacterial protein synthesis inhibitor that binds to the 30S subunit of the ribosome. It has been used as a second-line agent to treat gonorrheal infections, but with poor anti-tuberculosis activity.
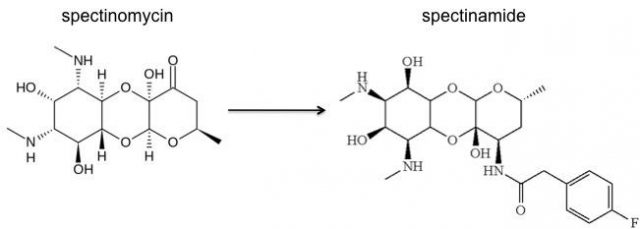
Taking the advantage that the crystal structure of spectinomycin bound to the ribosome has become available recently and using structure-based design, the authors generate spectinomycin analogs by the introduction of functional groups that have increased ribosomal target affinity. They synthetized a panel of spectinamides with good minimal inhibitory concentration activity specific to M. tuberculosis strains. The new compounds inhibited the mycobacterial ribosomal translation and were active against several M. tuberculosis strains, including drug-resistant strains, MDR and XDR tuberculosis clinical isolates, but not against an extensive panel of Gram-positive and Gram-negative bacteria.
In macrophage-resident M. tuberculosis, bacterial efflux pumps promote intracellular survival and induce intrinsic resistance to many antimicrobial agents as spectinomycin. These efflux systems help the bacteria to extrude antibiotics. However, the chemical variations on these new spectinamides overcome the efflux mechanisms in M. tuberculosis. This demonstrated that the structural modifications were able to evade the efflux pump, which is implicated in macrophage-induce drug tolerance.
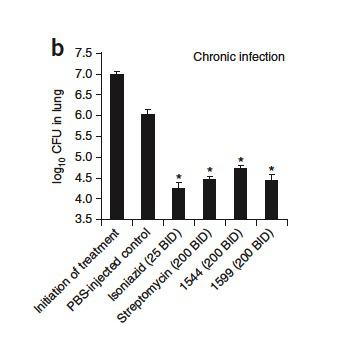
infected with M. tuberculosis treated with spectinamides (1544 and 1599), streptomycin and isoniazid. | Credit: Lee et al (2014)
In addition, spectinamides do not present detectable cytotoxicity against two mammalian cell lines (Vero and J774 cells) and do not inhibit mammalian translation, what reduce the potential for side effects. In vitro assays also showed that the spectinamides had low plasma protein-binding properties and were stable against hepatic microsomal metabolism, demonstrating favourable pharmacokinetic profiles. Finally, in vivo efficacy of spectinamides in four mouse tuberculosis aerosol infection models (acute infection, chronic tuberculosis, lethal infection and intrapulmonary nebulized drug delivery) was determined. These spectinamides were well tolerated, significantly reduced mycobacterial burden in lungs with no apparent toxicity and increased survival.
In summary, these synthetic modifications to classical antibiotics are potent bacterial ribosomal inhibitors that avoid efflux by M. tuberculosis to achieve excellent anti-tubercular efficacy in vivo. This study represents an example of new anti-tubercular drug class developed using semisynthetic approach to overcome resistant mechanisms. Next clinical assays about the in vivo efficacy of spectinamides are waiting. Meanwhile, a new hope against tuberculosis has been opened.
References
- Lee R.E., Hurdle J.G., Liu J., Bruhn D.F., Matt T., Scherman M.S., Vaddady P.K., Zheng Z., Qi J. & Akbergenov R. & (2014). Spectinamides: a new class of semisynthetic antituberculosis agents that overcome native drug efflux, Nature Medicine, 20 (2) 152-158. DOI: 10.1038/nm.3458 ↩
3 comments
[…] Uno de los problemas de la creciente resistencia a los antibióticos es que hay enfermedades, como la tuberculosis, tan mortal en el pasado, que están resurgiendo. Por ello son necesarios nuevos enfoques para combatirla y algunos paracen que son […]
[…] Gaixotasun batzuk antibiotikoei dioten erresistentziaren arazoetariko bat da, iraganean hilkorrak izan ziren tuberkulosia bezalako eritasunak berpiztu direla. Hortaz, honakoei aurre egiteko beharrezkoak dira ikuspuntu berriak eta badirudi etorkizun handikoak egon badaudela. Ignacio López Goñik kontatzen digu New hope against […]
[…] mappingignorance.org […]