Parasite plant check
Parasite plant check
Cuscuta (dodder) is a genus of about 200 species of obligate parasitic plants. They use airborne volatile organic compound cues to locate their host plants and then, they produce feeding structures named haustoria that penetrate into the vascular system of the host plants and suck their sap exploiting their carbohydrates, nutrients, and water. They also exchange macromolecules such as proteins and RNAs, as well as viruses, in a bidirectional manner. Some hosts are agricultural and horticultural species of high economic importance including potatoes and alfalfa and the damage by Cuscuta can be devastating, costing billions of dollars annually in crop losses across five continents, with particular impact in Africa 1.
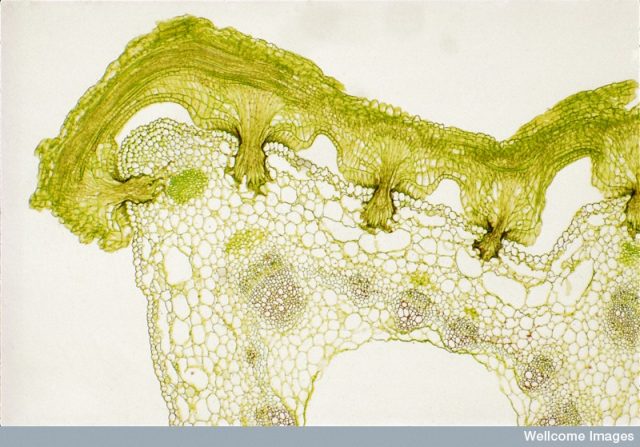
Light micrograph of a transverse section through dodder (Cuscuta sp) and its host stem, showing haustoria penetrating the host tissues. The section was stained with safranin and fast green. The safranin has stained the lignin in the xylem a reddish colour. Dodder has colourless twining stems and short-lived roots. It can seriously reduce the yield of crop plants.
The host plants defend against dodder using different mechanisms. Two pathways, using jasmonic acid and salicylic acid, are activated in response to attack by Cuscuta and different volatiles are synthesized that may interfere with the dodder’s system to locate and select hosts. However, we need better defense systems against dodder. Cuscuta nearly infect all plants, with the notable exception of tomato (Solanum lycopersicum) which can actively ward off the parasite attack.
Microbe-associated molecular patterns (MAMPs) are proteins associated with groups of pathogens, that are recognized by cells of the innate immune system. They are recognized by Toll-like receptors and other plasma-localized pattern recognition receptors (PRRs) in both plants and animals. These receptors initiate an effective immune response upon activation and thus plants are sometimes able to detect disease-causing microbes by the distinct MAMPs in their surface organizing a defense.

Up to now, it was unknown whether parasitic plants were also sensed as foes by a similar immune mechanism to that described for microorganisms. Volker Hegenauer and his group form the University of Tübingen (Germany) investigated the interaction between Cuscuta reflexa and cultivated tomato (Solanum lycopersicum). Tomato responds to C. reflexa extracts with a typical defense response after the detection of MAMPs like the production of stress-related phytohormone ethylene and reactive oxygen species. Authors purified and identified the Cuscuta factor using a procedure involving sequential separation steps. This factor appeared to be constitutively present in all parts of C. reflexa, including shoot tips, stems, haustoria, and at lower levels, in flowers. The tomato plant was hypersensitive to this extract, yet three other control plant species were not. The authors found in the host a surface immune receptor, renamed as CuRe1 (Cuscuta receptor 1), which recognizes the factor in the parasitic plant. CuRe1 is a small heat stable, potentially O-glycosylated and protease sensitive peptide, although its structure was not clearly identified. It is considered that the Cuscuta factor is associated or a structural component of the C. reflexa cell wall. Through this mechanism tomato plants deter attacks from the Cuscuta by detecting this peptide.
The factor is present in all Cuscuta species tested but is absent from related nonparasitic species Calystegia sepium or from unrelated parasitic plants 2. This contrasts with the current paradigm for MAMPs that indicates that they are widely conserved in a given class of organisms, in this case, plants themselves. This would imply an evident problem: If plants could respond to general plant factors, a predictable outcome might be autoimmunity (1) but in fact what is happening is that plants recognize the parasitic plants as non-self, similarly to perception of microbial or pest pathogens, without reacting to their own proteins 3. Two options may be that the factor may be genus-specific and it may be associated with a parasitic lifestyle or that it is a common cell wall protein with a specific posttranslational modification.
The CuRE1 is not the only resistance mechanism in the tomato since plants lacking CuRe1 were not susceptible to Cuscuta infection. In pathogenic microbia, haustoria are secretion sites for virulence proteins called effectors that promote pathogenesis but may be recognized by intracellular host immune complexes named R (resistance) proteins. We need to understand the nature of the Cuscuta factor and whether other parasitic plants are perceived by PRRs, whether parasitic plants inject effectors into their host as microbes do, whether R proteins from the host recognize these effectors and why these effectors are not active on their parasite plants of origin.
An important result is that CuRe1 is sufficient to confer responsiveness to the Cuscuta factor and increased resistance to parasitic C. reflexa when heterologously expressed in otherwise susceptible host plants. It is theoretically also possible to accelerate the engineering of receptors with novel ligand specificities. All this may offer future opportunity to engineer resistance to parasitic plants in crops.
References
- Ntoutakis V, Gimenez-Ibanez S (2016) Parasitic plants-A CuRe for what ails thee. Science 353(6298): 442-443. doi: 10.1126/science.aag3111 ↩
- Hegenauer V, Fürst U, Kaiser B, Smoker M, Zipfel C, Felix G, Stahl M, Albert M (2016) Detection of the plant parasite Cuscuta reflexa by a tomato cell surface receptor. Science 353(6298): 478-481. doi: 10.1126/science.aaf3919 ↩
- Hegenauer V, Fürst U, Kaiser B, Stahl M, Albert M, Smoker M, Zipfel C, G. Felix G (2016) The Tomato LRR-RLP CuRe1 detects molecular patterns of the parasitic plant Cuscuta reflexa. 2016 International Society for Molecular Plant-Microbe Interactions Congress. ↩
2 comments
[…] Los parásitos son un mundo fascinante, por lo ingeniesos que son sus mecanismo para explotar las vulnerabilidades de sus huéspedes. También ocurre en las plantas. Pero, de vez en cuando, se encuentran mecanismos de defensa. JR Alonso en Parasite […]
[…] Los parásitos son un mundo fascinante, por lo ingeniesos que son sus mecanismo para explotar las vulnerabilidades de sus huéspedes. También ocurre en las plantas. Pero, de vez en cuando, se encuentran mecanismos de defensa. JR Alonso en Parasite […]