A new pathway to avoid macrophage pathogenic infections
A new pathway to avoid macrophage pathogenic infections
During the Cold War the competition between both sides, Russia and the United States of America, caused a quick development of military and spacial technology: while the Soviet Union was the first country responsible for an orbital launch (Sputnik 1), the first nation to ever send a man to space and pioneered spacewalks, the U. S. A. put a man on the moon. The history of humanity is full of examples of arm races and technology wars. Swords conditioned the production of armours and siege weapons evolved hand in hand with fortresses, which became nearly outdated when gunpowder arrived.
The history of life itself is also conditioned by diverse arm races. Some of them are quite evident: cheetah and gazelles became engaged in a literal race that resulted in both species increasing progressively their manoeuvrability. But there are also arm races inside the organisms; molecular and cellular races. The study of Dr. Valledor´s group is focused in an example of these competitions: the interrelation between macrophages and certain pathogens.
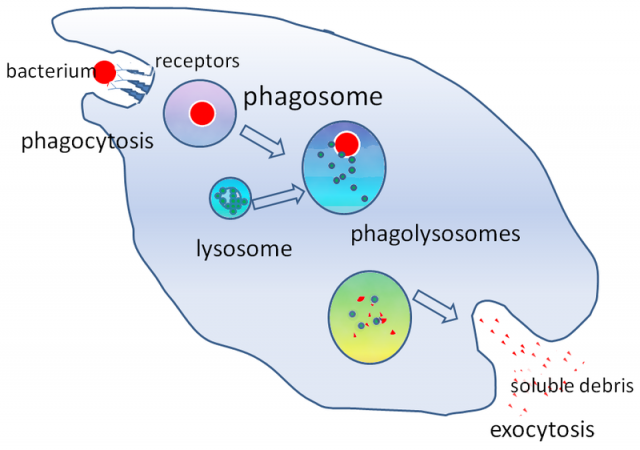
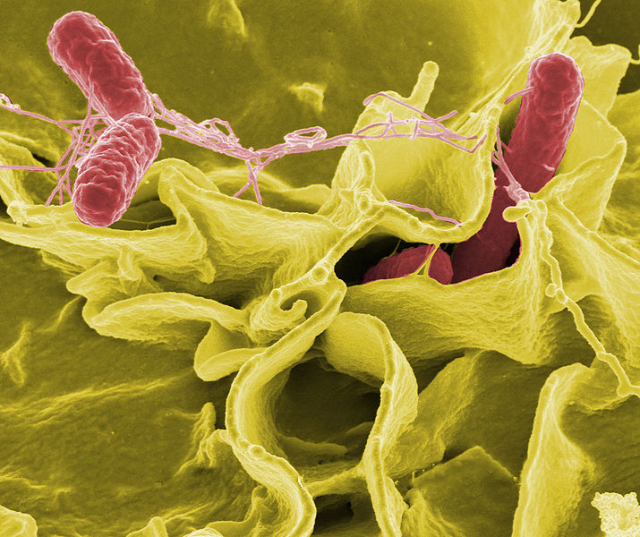
Macrophages are cells that play a significant role in the immune system, specialized in detecting and eliminating strange bodies (literally swallowing them or phagociting them). However, some bacteria, like some strains of Salmonella among others, not only are capable of resisting phagocytosis but surprisingly they are also adapted to survive and reproduce inside those cells.
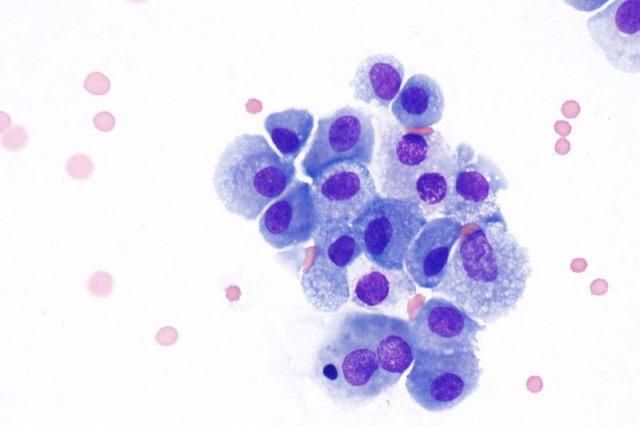
They use macrophages as a niche, hiding from the entire immune system; they make macrophages their home, using them as microscopic Trojan horses or analogical Trojan computer programs (if we wish to upgrade the myth). The study of the molecular mechanisms regulating this “trojan activity” will allow to identify possible treatments to fight these attacks. Dr. Valledor’s group is centred in the study of the role of nuclear receptor LXR in such infections. But why LXR?
LXR (Liver X Receptor) is a nuclear receptor that was initially associated with lipid metabolism. Nevertheless, its involvement in several processes and cells of the defence system has also been proved. More precisely, it has been observed that genetically modified mice lacking LXR showed an increased susceptibility to infections by different bacteria. Besides, other studies have shown how LXR activation protected macrophages from being killed by other bacteria like Salmonella itself.
Dr. Valledor’s group shows 1 that treatment with molecules that activate LXR (receptor’s agonists) protects macrophages from Salmonella infection, that is,they ascertained a smaller number of infected macrophages. This does not take place when the cells treated with the agonists do not have LXR (because it was previously removed using genetic engineering). Therefore, the action of these molecules is exerted through this nuclear receptor and the effect was observed both in cell cultures and mice.
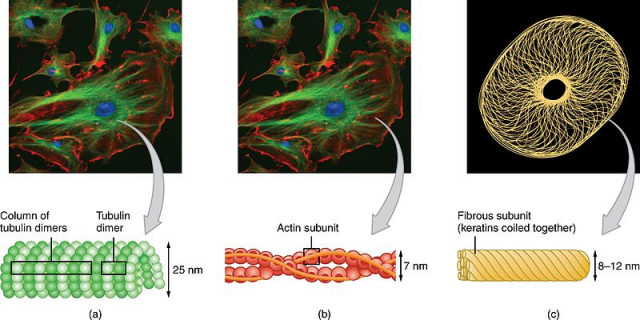
Salmonella infection commonly alters the shape of the macrophages and when these were treated with the LXR agonists the original shape was recovered.
This work deeply explores the molecular mechanisms implicated in the change of shape. It is proved that LXR reduces intracellular levels of a small molecule, NAD+, and this is mediated by the induction of a protein known as CD38. When CD38 is induced and NAD+ levels decrease, macrophage morphology remains intact because their cytoeskeleton, this is, the network of tubules and filaments that give the cell its shape and mechanical resistance, cannot be modified by the pathogenic bacteria.
In order to test viable pharmacological applications, mice were infected with Salmonella typhimurium and some of them were treated with an activator (agonist) of LXR. This treatment ameliorated the mice response to the infection: they did not lose so much weight and they presented moderate clinical symptoms. Moreover, the treatment clearly diminished the infection in the spleen and the liver. Transgenic mice lacking LXR or CD38, however, did not show any sign of improvement, so it can be deduced that the mechanism of LXR activation needs both the gene itself as well as the CD38 protein to operate properly.
Conclusively, this work identifies a novel pathway that may lead to effective pharmacological treatments to cure infections caused by organisms that use macrophages as Trojan horses.
References
- J. Matalonga et al (2017) The Nuclear Receptor LXR Limits Bacterial Infection of Host Macrophages through a Mechanism that Impacts Cellular NAD Metabolism Cell Reports doi: 10.1016/j.celrep.2017.01.007 ↩
1 comment
[…] Check our article in Mapping Ignorance: A new pathway to avoid macrophage pathogenic infections. […]