Plasmonic nanocrystals-cellulose hybrid
Plasmonic nanocrystals-cellulose hybrid
When we consider the variety of possible wonder applications of plasmonic nanoparticles we usually forget a key aspect: how these laboratory results can be transformed into something really usable in everyday life. For that to occur some not-that-simple problems must be resolved first.
For example, plasmonic nanoparticles exhibit excellent light-harvesting properties in the visible spectral range. Wonderful, we think, this could be a way for the conversion of light into useful chemical fuel, just like plants do. The problem is that, in order to achieve this, nanoparticles need to be, well, nanoparticles. If nanoparticles, fabricated in liquid phase, aggregate, they are no longer nanoparticles, but microparticles or miliparticles, or whatever, you get the idea. So, we have to maintain nanoparticles as such: enter the colloidal solution.
In order to maintain the nanoparticles as a colloid, we need to stabilize them, usually using surface ligands. Now, we have nanoparticles but not the promised light-harvesting properties, as ligands inhibit their photochemical performance, due to the insulating molecular shell hindering the carrier transport.
How to solve this? Ideally, we would need an easy to manipulate, cheap, abundantly available, inert from the plasmonic point of view material that we could use as a scaffold for our nanoparticles. Does something like this exist ? Now, a team of researchers think 1 they have found a candidate: cellulose.
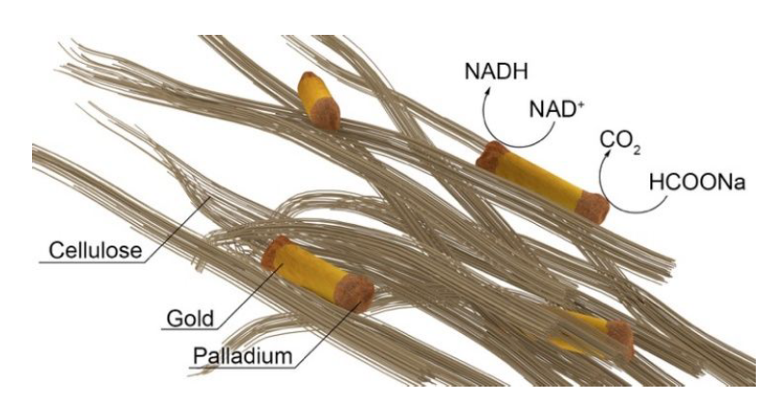
Of course, the strategy of immobilizing nanocrystals on solid substrates is not new. Several types of substrates have been proposed so far, including TiO2, CeO2, ZrO2, Al2O3, SiO2, MoS2, glass or, why not, graphene. The electronic properties of those materials, however, as the surface ligands before, also alter the dynamics of carriers flow.
Cellulose is an excellent scaffold candidate for the plasmonic catalyst because of its availability, chemical robustness, and high-surface area. Interestingly, the combination of cellulose with semiconducting nanoparticles has been shown to produce an efficient photocatalyst for the generation of hydrogen or the degradation of an organic dye when irradiated with visible light.
On the other hand, the shape anisotropy of cellulose fibers has been exploited in a templated self-assembly of plasmonic nanoparticles leading to chiral optical properties, or serving as a substrate for plasmon-based biosensing.
The researchers show now that a combination of cellulose with metal nanocrystals (Pd-coated gold nanorods) renders a composite with photocatalytic activity in the dehydrogenation of sodium formate and the simultaneous reduction of NAD+to NADH under visible-infrared light irradiation.
The excess of cationic surfactants on the surface of the particles likecetyltrimethylammonium bromide (CTAB) alters the metal-cellulose interactions. The fabrication of a functional composite was possible by the rational screening of the concentrations of CTAB, nanoparticles, and cellulose in the mix, thus obtaining processable materials in the form of gels, viscous solutions, two-phase systems or stable colloidal solutions. The initial optical and photocatalytic properties of the palladium-gold nanocrystals were kept by fine-tuning the metal-cellulose interactions, even after the removal of the CTAB surfactant.
The resulting functional composite was implemented in a bioinspired photocatalytic process involving dehydrogenation of sodium formate and simultaneous photoregeneration of cofactor molecules (NADH, nicotinamide adenine dinucleotide) using visible light as an energy source.
The functionality of this new plasmonic nanocrystals-cellulose hybrid system brings processable macroscopic materials closer to a reality. Including the use of 3D printing technology.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may be copied verbatim or almost verbatim from the referenced research paper.
References
- Nina Tarnowicz-Staniak, Silvia Vázquez-Díaz, Valeri Pavlov, Katarzyna Matczyszyn, and Marek Grzelczak (2020) Cellulose as an Inert Scaffold in Plasmon-Assisted Photoregeneration of Cofactor Molecules ACS Appl. Mater. Interfaces doi:10.1021/acsami.9b21556 ↩