PIM1 Activation in T-ALL: A Therapeutic Vulnerability
PIM1 Activation in T-ALL: A Therapeutic Vulnerability
Author: Marta Irigoyen is a postdoctoral researcher at CIC bioGUNE
T-cell acute lymphoblastic leukemia (T-ALL) and T-cell acute lymphoblastic lymphoma (T-LBL) are aggressive hematological malignancies that arise from abnormal activation of oncogenes and/or inactivation of tumor-suppressor genes, followed by a differentiation arrest and uncontrolled clonal expansion of immature thymocytes 1. Proviral integration site for Moloney-murine leukemia 1 (PIM1) is a known JAK-STAT target gene that recently emerged as a therapeutic target for the treatment of T-ALL and T-LBL2. However, recent work suggested that activation of JAK-STAT signaling in T-ALL could also be achieved through non–cell-autonomous mechanisms, such as stimulation by interleukin 7 (IL7) 3 This cell-extrinsic mechanism of JAK-STAT pathway activation suggests that the fraction of T-ALL and T-LBL patients that might benefit from in vivo PIM-inhibitor therapy could be more substantial than originally anticipated. Notably, previous research has shown that glucocorticoids, one of the core components of T-ALL/T-LBL induction therapy, can directly induce Il7r expression (the receptor of the interleukin 7) in murine and human T cells 4. Therefore, glucocorticoids could potentially drive therapy-induced and non–cell-autonomous activation of the JAK-STAT pathway, eventually leading to downstream PIM1 activation. Based on this, the authors of the present study hypothesized that T-ALL/T-LBL cells that evade induction therapy may also be characterized by elevated PIM1 expression 5.
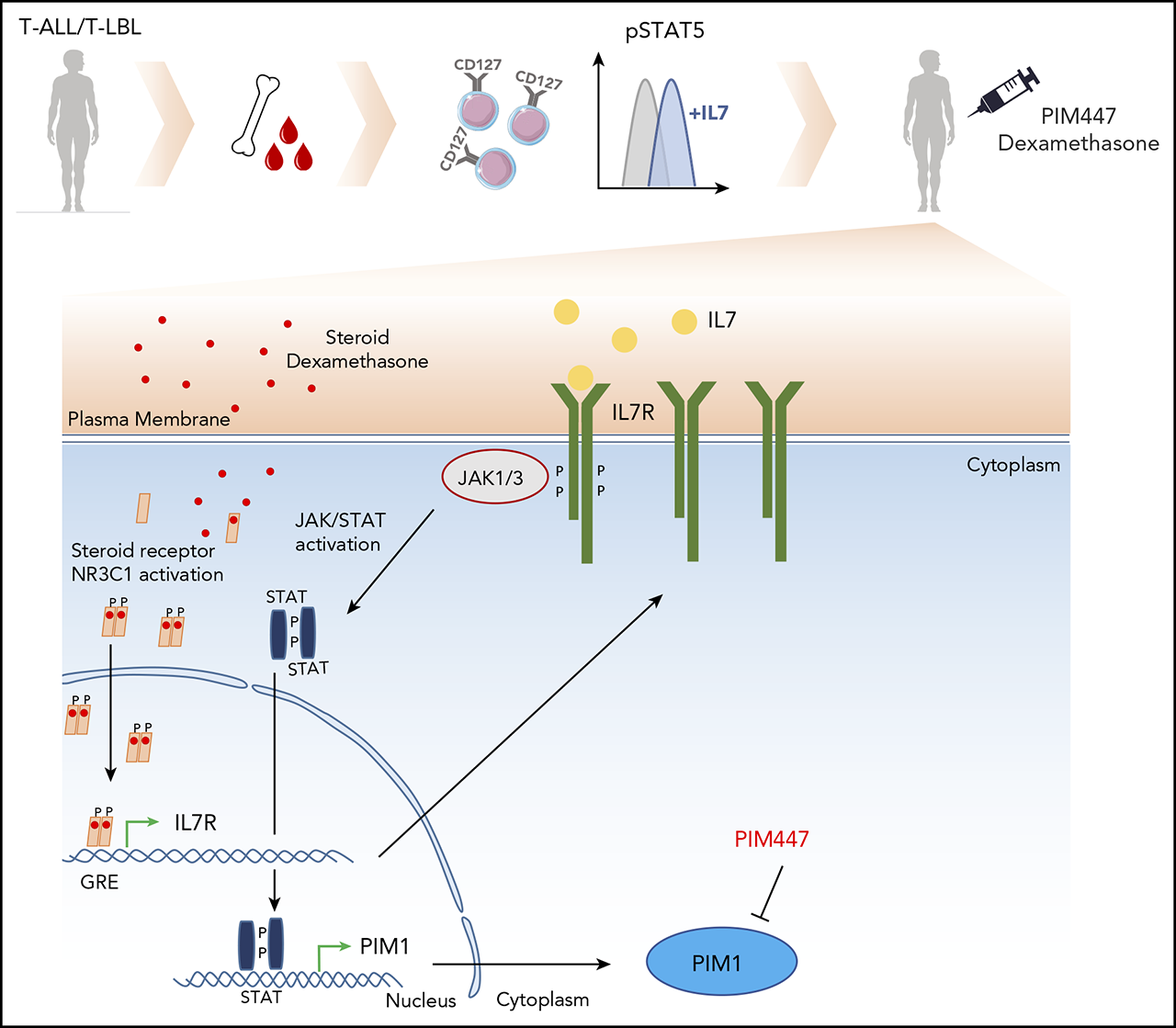
Previous studies have shown that a subset of T-ALL patients can activate JAK-STAT signaling in response to exogenous IL7. However, the specific genetic characteristics of T-ALL/T-LBL patients exhibiting this IL7 responsiveness remain largely unknown. To investigate this, the authors analyzed pSTAT5 levels following IL7 stimulation in a set of genetically well-characterized PDX samples derived from 7 pediatric T-ALL and 4 T-LBL patient cases. From these 11 PDX samples, 4 T-ALL and 2 T-LBL cases showed IL7-induced pSTAT5 induction, which was not the case for the remaining 5 T-ALL/T-LBL samples. Notably, IL7-responsive T-ALL/T-LBL samples were predominantly characterized by higher CD127 expression compared to non-responders. As expected, IL7 stimulation resulted in PIM1 upregulation in responsive T-ALL/T-LBL patient samples, suggesting that CD127+ T-ALL/T-LBLs might also benefit from PIM inhibition.
To explore this, the authors conducted in vivo testing of PIM inhibitors using a T-ALL xenograft model known to upregulate PIM1 expression upon IL7 stimulation. They observed that both PIM inhibitors were able to reduce the leukemic burden. PIM1 overexpression in T-ALL/T-LBL can be caused by genomic translocations involving T-cell receptor β (TCRβ) or activating mutations targeting the IL7R/JAK/STAT signaling pathway. However, as previously shown, PIM1 can also become activated through stimulation by exogenous IL7 in CD127+ T-ALL/T-LBL. To determine whether these distinct mechanisms of PIM1 activation are mutually exclusive, the authors stimulated leukemic cells that already express high levels of PIM1 due to a JAK1 mutation. Upon stimulation, these cells exhibited clear pSTAT5 induction accompanied by a significant upregulation of PIM1. Similarly, IL7 stimulation of leukemic cells also led to pSTAT5 activation and concurrent PIM1 upregulation.
As already mentioned, glucocorticoids are core components of T-ALL treatment 6 which bind to the glucocorticoid receptor (NR3C1) inducing apoptosis in lymphoid cancers. Interestingly, previous research showed that glucocorticoids are able to bind an enhancer of the IL7RA locus, thereby upregulating IL7RA expression in thymocytes 7. Given this, the authors evaluated IL7RA expression in their panel of 11 T-ALL/T-LBL PDX samples upon dexamethasone (a common glucocorticoid used in T-ALL treatment) treatment. The analysis confirmed that glucocorticoids can induce IL7RA expression in most leukemic samples. Finally, they wanted to determine whether glucocorticoid-induced IL7RA activation would also result in JAK-STAT pathway activation with concomitant upregulation of PIM1. Indeed, dexamethasone treatment of PDX samples in combination with IL7 was able to cause a significant upregulation of PIM1 expression along the JAK-STAT axis in most T-ALL/T-LBL PDX samples. To determine whether this mechanism of glucocorticoid-induced PIM1 activation would also take place when leukemic cells are exposed to induction therapy in an in vivo IL7-producing microenvironment, authors performed additional T-ALL/T-LBL xenograft experiments in which they treated mice for 1 week with a chemotherapeutic cocktail or vehicle control. After 1 week, both control and chemotherapy-treated mice were euthanized and leukemic cells were isolated from the bone marrow.
Notably, quantitative RT-qPCR revealed a significant upregulation of PIM1 expression in residual leukemic blasts as compared with controls in four of the analyzed samples. This result confirmed that the induction chemotherapy can trigger in vivo JAK-STAT pathway activation in human T-ALL and T-LBL. In agreement with previous results showing that the in vivo combination therapy of a PIM inhibitor with glucocorticoids significantly prolonged animal survival using a PDX model of a T-LBL case 8, the authors decided to include PDX cells from T-LBL patient sample demonstrating a dual activation of both cytokine and therapy-induced PIM1. Therefore, the previously published synergistic effects of this combination therapy were probably mediated by a combination of both cell-intrinsic and cell-extrinsic effects, which ultimately resulted in robust in vivo PIM1 activation.
To further investigate, the authors assessed the combined effect of the PIM inhibitor PIM447 and dexamethasone on PDX cells derived from both IL7-responsive and non-responsive T-ALL/T-LBL samples, in the presence of exogenous IL7. Interestingly, synergism was observed in the IL7-responder group, whereas no beneficial effect was seen in IL7 non-responders. Next, they evaluated this combination therapy in vivo using and observed a significant improvement in survival for the PIM447/dexamethasone combination as compared with both monotherapy treatments. Finally, they assessed the combination of PIM447 with chemotherapy in vivo using another sample that lacked cell-intrinsic genetic abnormalities targeting PIM1 or the IL7R pathway, but had the ability to activate PIM1 upon IL7 stimulation and induction chemotherapy. Notably, this analysis also revealed a significant improvement in survival in the PIM447/VXL combination group as compared with monotherapy.
Altogether, their study shows that cytokine- and therapy-induced PIM1 activation can be therapeutically targeted by the pan-PIM inhibitor PIM447 in human T-ALL/T-LBL. In addition, their work suggests that IL7 responsiveness in CD127+ T-ALL/T-LBL could serve as a valuable biomarker to identify patients that might benefit from PIM inhibition during induction chemotherapy.
References
- Van Vlierberghe P, Pieters R, Beverloo HB, Meijerink JP. (2008) Molecular-genetic insights in paediatric T-cell acute lymphoblastic leukaemia. Br J Haematol. 2008;143:153-168. PMID: 18691165DOI: 10.111/j.1365-2141.2008.07314.x. ↩
- La Starza R, Messina M, Gianfelici V, Pierini V, Matteucci C, Pierini T, Limongi MZ, Vitale A et al. (2018) High PIM1 expression is a biomarker of T-cell acute lymphoblastic leukemia with JAK/STAT activation or t(6;7)(p21;q34)/TRB@-PIM1 rearrangement. Leukemia. 2018;32:1807-1810. PMID: 29479063 DOI: 10.1038/s41375-018-0031-2 ↩
- Delgado-Martin C, Meyer LK, Huang BJ, Shimano KA, Zinter MS, Nguyen JV, Smith GA, Taunton J et al. (2017) JAK/STAT pathway inhibition overcomes IL7-induced glucocorticoid resistance in a subset of human T-cell acute lymphoblastic leukemias. Leukemia. 2017;31:2568-2576. PMID: 28484265 DOI: 10.1038/leu.2017.136. ↩
- Franchimont D, Galon J, Vacchio MS, Fan S, Visconti R, Frucht DM, Geenen V, Chrousos GP et al (2002) Positive effects of glucocorticoids on T cell function by up-regulation of IL-7 receptor alpha. J Immunol. 2002;168:2212-2218. PMID: 11859107DOI: 10.4049/jimmunol.168.5.2212. ↩
- De Smedt R, Morscio J, Reunes L, Roels J, Bardelli V, Lintermans B, Van Loocke W, Almeida A et al (2020) Targeting cytokine- and therapy-induced PIM1 activation in preclinical models of T-cell acute lymphoblastic leukemia and lymphoma. Blood. 2020 May 7;135:1685-1695. PMID: 32315407 DOI: 10.1182/blood.2019003880. ↩
- Inaba H, Pui CH. (2010) Glucocorticoid use in acute lymphoblastic leukaemia. Lancet Oncol. 2010;11:1096-1106. PMID: 20947430 DOI: 10.1016/S1470-2045(10)70114-5. ↩
- DeKoter RP, Schweitzer BL, Kamath MB, Jones D, Tagoh H, Bonifer C, Hildeman DA, Huang KJ. (2007) Regulation of the interleukin-7 receptor alpha promoter by the Ets transcription factors PU.1 and GA-binding protein in developing B cells. J Biol Chem. 2007;282:14194-14204. PMID: 17392277 DOI: 10.1074/jbc.M700377200. ↩
- De Smedt R, Peirs S, Morscio J, Matthijssens F, Roels J, Reunes L, Lintermans B, Goossens S et al (2019). Pre-clinical evaluation of second generation PIM inhibitors for the treatment of T-cell acute lymphoblastic leukemia and lymphoma. Haematologica. 2019;104:e17-e20. PMID: 30076176 DOI: 10.3324/haematol.2018.199257. ↩