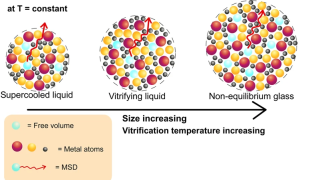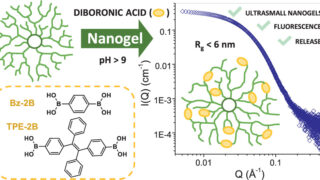
Molecular origami: Crafting ultrasmall nanogels through intramolecular architecture
Nanogels are among the most interesting examples of how chemists can build useful structures by working at the scale of single molecules. They are tiny soft particles made from polymers, long chain-like molecules, that are linked together into small three-dimensional networks. Because they can hold water and trap other molecules inside, nanogels are especially attractive […]