Touch me, please

Autism is a highly prevalent spectrum disorder of the neurodevelopment. It is characterized by impaired social interaction, alterations in verbal and non-verbal communication, and restricted and repetitive behaviors. Considering these main differences, it had been difficult to have good animal models since these interactions were initially defined for humans but scientists are nowadays developing different strains of rodents that have some behaviors, some genes or some neural anomalies similar to those from persons affected by autism.
Most people think that autism is a disorder of the brain but there are evidence relating it with the organism as a whole. As an example, babies who later will develop autism are larger than their unaffected siblings and the reason is presently unknown. In addition, autism spectrum disorders (ASDs) do clearly influence sensory function. Many persons with autism have hypo- or hypersensitivity for one or more senses and, in fact, 95% of individuals diagnosed with ASDs also exhibit aberrant reactivity to sensory inputs. ASD patients and the associated animal models of these disorders often have visual, auditory, olfactory, gustatory, or tactile abnormalities. Therefore, impairments in sensory systems during critical developmental windows may contribute to abnormal language acquisition, cognition, anxiety, and abnormal social behaviors, typical characteristics of persons with autism.
A majority of persons with autism (60.9%) exhibit altered tactile sensitivity in both glabrous (smooth) and hairy skin as well as an augmented sensitivity to vibration and thermal pain. They tend to be very wary of social touch (like a hug or a handshake) and can be overwhelmed by certain textures. Since childhood tactile experiences are critical for the acquisition of normal social and communication behaviors, a relation between tactile disruption and ASDs can be hypothesized.
Although we usually relate autism with the brain, a recent study published in Cell 1 suggests that at least some aspects of the ASD —including touch perception, anxiety, and social oddities — are linked to defects not in the central nervous system but in the peripheral nervous system. The sensation of touch starts in skin receptors and travels along nerves that connect into the central nervous system. Up to now, we centered just on that final part of the pathway.
The investigators studied in genetically engineered mice the effects of gene mutations known to be associated with ASD in humans. One of the chosen genes was Mecp2, which causes Rett syndrome, a disorder where affected persons demonstrate autistic symptoms. Other analyzed genes were Gabrb3, Shank3, and Fmr1, that are involved in synaptic function and neuron connectivity and are related to ASD-like behaviors as well. The research team designed useful approaches to studying the effects of the mutations. They introduced mutations that silenced those genes associated with ASD, adding them in a way that restricted their expression both temporarily (development vs. adulthood) and spatially (central vs. peripheral).
Mutant mice and control littermates were subjected to tactile-based tasks to assess mechanosensory behaviors and sensitivity. The researchers developed a texture-specific novel object recognition test (NORT). The deficits were specific for textured NORT, and not a general lack of novelty-seeking behavior, as mutant mice performed comparably to control mice on a control NORT in which objects differed in color and shape, but not in texture. In addition, the Mecp2 mutant mice were more sensitive to light touch, a small puff air on air on their backs startled the rodents more than normal mice.
Mecp2 and Gabrb3 function in somatosensory neurons for normal tactile behaviors. Sensory neuron deletion of Mecp2 led to a decrease in Gabrb3 in the dorsal horn, and Gabrb3 expression in sensory neurons was required for tactile sensitivity. The main result was that these mutations were both necessary and sufficient for creating mice with an abnormal hypersensitivity to touch. 2 Interestingly, tactile alterations after Mecp2 or Gabrb3 deletion in somatosensory neurons during development, but not in adulthood, caused social interaction deficits and anxiety. Autism may, therefore, arise, in part, from an altered sense of touch.
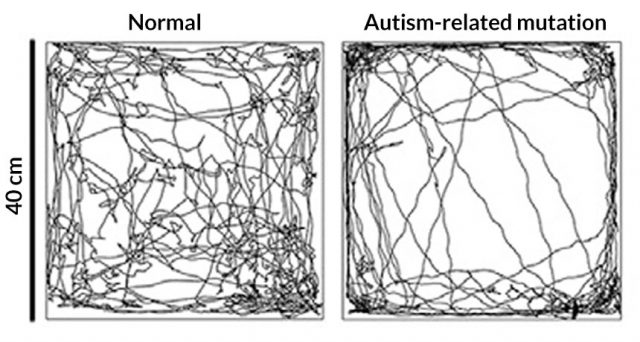
The mice also displayed autism-like behaviors beyond touch. Animals with the defective Mecp2 but not in brain cells were also more anxious and less social, traits generally attributed to the central nervous system. When given the alternative to interact with another mouse or an object, the mutant mice spent as much time with the object as with the other animal, unlike normal mice, which significantly prefer a living companion. Tests also found differences in anxiety levels. Normal mice explore the entirety of an open area whereas mutant mice preferred to stay on the edges, a behavior suggesting heightened anxiety. 3
A clear evidence for peripheral involvement was obtained using a viral therapy to correct mutations. Systemic viral replacement of Mecp2 in Mecp2 hemizygous male mice effectively rescued behavioral deficits relevant to some ASD phenotypes. However, when the Mecp2 was delivered used the same viral tool but only intracranially, it only mildly improved the altered behaviors.
Another interesting similarity was that Mecp2 mutant mice nerves had low levels of GABA receptors. Low GABA levels in the brain have previously been linked to autism, but the new finding opens up an unexpected treatment possibility: a drug that restores GABA function in the periphery.
To further evaluate social interactions, mice were subjected to a tube dominance test, which allowed the researchers to assess social approach/avoidance behavior, as well as evaluate dominance hierarchies in mice. These tests have been previously used to assess social impairments in ASD mouse models. Mecp2 and Gabrb3 mutant mice exhibit decreased social dominance and aggression, as they lost the majority of matches against control mice. These results were consistent with previous reports indicating that Mecp2-null mice exhibit increased escape and avoidance behaviors in a predator-threat stimulus test. In contrast, mice in which either Mecp2 or Gabrb3 was deleted in somatosensory neurons in adulthood demonstrated increased dominance, as these mice won the majority of their matches. All considered, these observations suggest that there is a developmental need of Mecp2 and Gabrb3 in primary somatosensory neurons for the acquisition of certain cognitive and social behaviors in mice.
The behavioral results observed in this study were reminiscent of human, non-human primate, and rodent tactile deprivation studies. These studies have concluded that early life experiences and tactile stimulation during the development are crucial for proper brain organization, cognition, and social life in adults. A sad additional evidence is that children who are reared in state institutions with bad quality care and low physical handling exhibited deficits in cognitive function, delayed or impaired language acquisition and social interactions. This results in an increased incidence of anxiety and psychiatric disorders.
References
- Orefice LL, Zimmerman AL, Chirila AM, Sleboda SJ, Head JP, Ginty DD (2016) Peripheral Mechanosensory Neuron Dysfunction Underlies Tactile and Behavioral Deficits in Mouse Models of ASDs. Cell doi: 10.1016/j.cell.2016.05.033 ↩
- Cell Press. “Autism is not just a disorder of the brain, mouse study suggests.” ScienceDaily. ScienceDaily, 9 June 2016. ↩
- Burrell T (2016) Autism may stem —in part— from a disordered sense of touch. Science Magazine news ↩