Penta-graphene: to be or not to be
Penta-graphene: to be or not to be
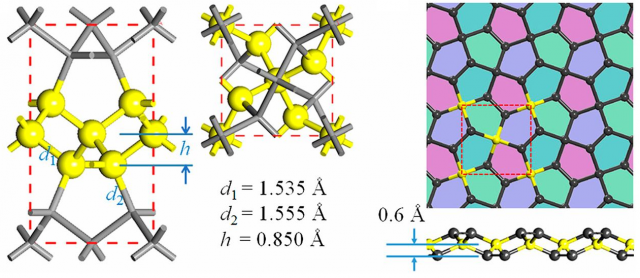
Graphene is a two-dimensional allotrope of carbon made of hexagons. In February 2015, penta-graphene, only made of pentagons, was theoretically proposed as another two-dimensional allotrope of carbon 1. Apparently, it is dynamically, thermally, and mechanically stable, exhibiting a large band gap (graphene is gapless). However, an exhaustive experimental search has been inconclusive, resulting in doubts on its experimental attainability 2. Recent results suggest that penta-graphene and graphene can be considered as crystallographic reorientations of the same material, rather than distinct allotropes 3. In fact, this can be the case for other theoretically proposed carbon allotropes, like ph-graphene 4, having a penta-hexagonal structure.
The main drawback for graphene as a transistor material is the absence of band gap for switch on-off operation. Penta-graphene solves this problem having an intrinsic band gap as large as 3.25 eV. It is composed entirely of carbon pentagons, resembling the Cairo pentagonal tiling, alternating between sp3– and sp2-hybridized carbon atoms [1]; let’s recall that in graphene all the carbon atoms are sp2-coordinated and in diamond all are sp3-coordinated. Ab initio molecular dynamics (AIMD) simulations predicts a high thermal stability of penta-graphene even at temperatures as high as 1000 K.
The successful synthesis of penta-graphene will trigger considerable interest in a large number of practical applications. For the fabrication of penta-graphene, Shunhong Zhang, from Peking University, Beijing, China, and colleagues [1] have proposed the exfoliation from T12-carbon, another theoretically proposed tetragonal allotrope of carbon, but three-dimensional with 12 atoms per cell 5. However, the experimental efforts have been inconclusive; penta-graphene is energetically metastable compared with graphene.
A set of criteria to evaluate whether theoretically proposed carbon allotropes may be experimentally isolated has recently been introduced by the late Sir Harold W. Kroto (1939–2016), Florida State University, FL, USA, and several colleagues [2]. The stable allotropes must occupy deep and isolated potential wells in the energetic landscape, whose large basin of attraction avoids the “funneling” toward more stable structural forms. The application of these criteria to penta-graphene demonstrates with the aid of density functional calculations that it will not be experimentally attainable. In fact, penta-graphene is 0.761 eV per atom less stable than graphene. Hence, it may transform directly to graphene by means of a sequence of bond rotations and bond breakages.
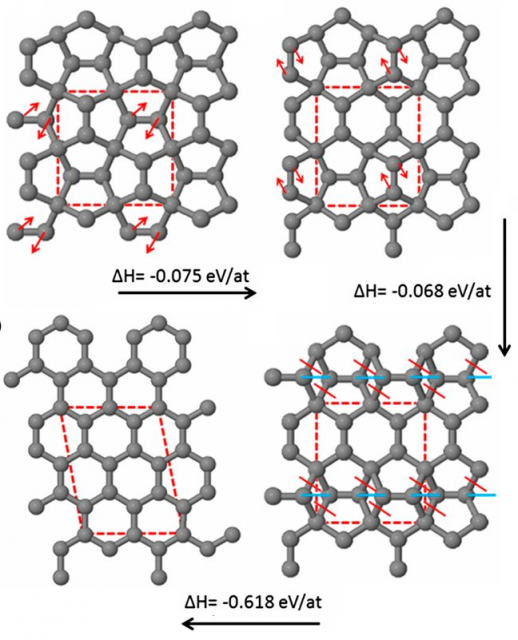
The emergence of one material (graphene) from another metastable form (penta-graphene) is a new paradigm in nanomaterials [3]. The transition from penta-graphene to graphene has been studied in detail by means of full atomistic molecular dynamics (MD) by Steven W. Cranford, Northeastern University, Boston, MA, USA. His paper is entitled “six is less than five” because the hexagonal 6-ring structure of graphene represents an energy minimum less than the pentagonal 5-ring structure of penta-graphene.
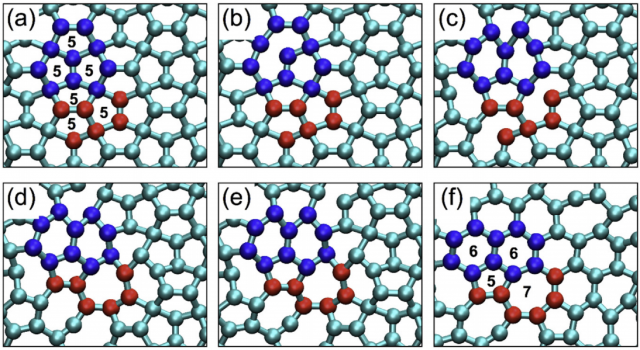
During the phase transformation of penta-graphene into graphene, stable intermediate forms with a mixture of pentagons and hexagons are observed. The basic step is the reform of three adjacent pentagons (10 carbon atoms) into two adjacent hexagons, accompanied by a (5,7)-defect. In this process energy is released, but penta-graphene does not fracture. Hence, this structural transformation propagates across the 2D sheet by repetition of the basic step. The process ends when the original penta-graphene is effectively graphene (with some intrinsic defects). From this perspective, penta-graphene can be considered as an extremely “defective” graphene, i.e., both materials are crystallographic reorientations of the same allotrope rather than distinct ones.
Currently, a large number of new carbon allotropes composed of pentagons and hexagons are being proposed from first-principles calculations. For example, ph-graphene, which is energetically more favorable than the penta-graphene [4]. These materials are aesthetically fascinating, but they cannot be considered carbon allotropes unless they pass the criteria discovered by Kroto and colleagues. In Science, as Ophelia tells to Hamlet, “could beauty, my lord, have better commerce than with honesty?”
References
- Shunhong Zhang et al., “Penta-graphene: A new carbon allotrope,” PNAS 112: 2372–2377 (Feb 2015), doi: 10.1073/pnas.1416591112. ↩
- Christopher P. Ewels et al., “Predicting experimentally stable allotropes: Instability of penta-graphene,” PANS 112: 15609–15612 (Dec 2015), doi: 10.1073/pnas.1520402112. ↩
- Steven W. Cranford, “When is 6 less than 5? Penta- to hexa-graphene transition,” Carbon 96: 421–428 (Jan 2016), doi: 10.1016/j.carbon.2015.09.092. ↩
- Xiaoming Zhang et al., “Prediction of an ultrasoft graphene allotrope with Dirac cones,” Carbon 105: 323–329 (Aug 2016), doi: 10.1016/j.carbon.2016.04.058. ↩
- Zhisheng Zhao et al.,Tetragonal Allotrope of Group 14 Elements,” Journal of the American Chemical Society 134: 12362–12365 (Jul 2012), doi: 10.1021/ja304380p. ↩
3 comments
[…] leer mi última contribución “Penta-graphene: to be or not to be,” Mapping Ignorance, 25 Jul 2016, sobre el pentagrafeno (propuesto a principios de 2015) y la posibilidad de que no sea un nuevo […]
[…] El pentagrafeno, propuesto a principios de 2015, podría no ser un nuevo alótropo del carbono, sino una reorentación cristalográfica del grafeno. Francisco R. Villatoro en Penta-graphene: to be or not to be. […]
[…] El pentagrafeno, propuesto a principios de 2015, podría no ser un nuevo alótropo del carbono, sino una reorentación cristalográfica del grafeno. Francisco R. Villatoro en Penta-graphene: to be or not to be. […]