Ruthenium nitrosyl complexes: a useful kind of molecular photoswitches
Ruthenium nitrosyl complexes: a useful kind of molecular photoswitches
Author: Francesco Talotta is a Ph.D. student (ITN-EJD-TCCM) at Université Toulouse III
A molecular switch consists in a molecular device able to change its physical or chemical properties (as its color for example) in a well definite way. In the best cases changing can be fully controlled, normally by an external chemical or physical factor, like light irradiation or temperature. The process of vision is arguably the most fantastic among nature’s numerous systems that can be triggered by a switching process at the molecular level. In this moment when you’re reading this sentence, a large collection of molecular switches are changing their state to make this possible in you eyes! In the ongoing quest for nano-devices and molecular machines, the design of molecular switching elements integrated with a variety of functions represents a very important research field and a formidable challenge at the same time. For this reason, in past years many efforts have been made in design, investigation and production of devices capable of act as switches at molecular level. Research covers a wide range of frontiers in science from molecular computing to light sensors, display and smart materials, stimulated in particularly by prospects of memory elements as small as the single molecule. Molecular switches can indeed store informations through binary digits 0 and 1.
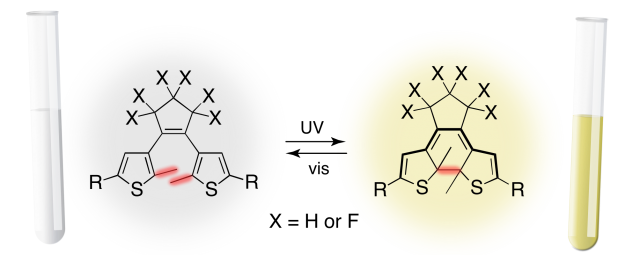
One of the first examples of molecular systems like this is represented by the DTE molecule, synthesized in the late 1980’s. Experimental evidences 1 pointed out how an aqueous DTE solution changes its color from transparent to yellow upon ultraviolet irradiation (UV), as shown in Figure 1. At molecular level reaction involves a changing in DTE molecular structure, with closure of the central ring. A new isomer with different optical properties is thus generated by light irradiation (right molecule in Figure 1).
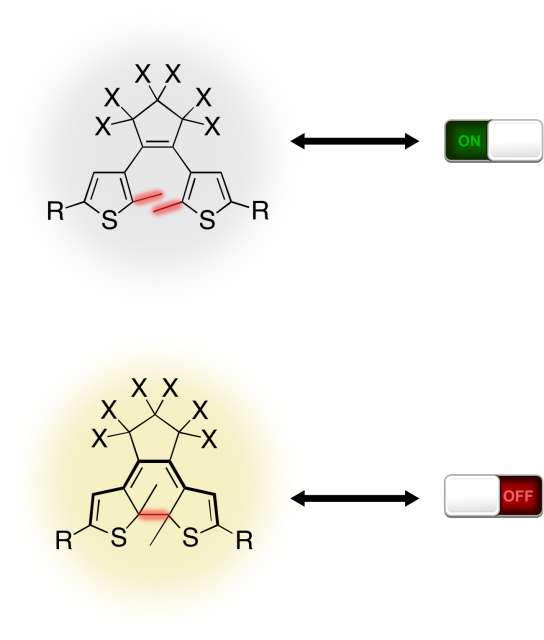
Figure 2. The DTE isomers as a molecular switch.
The closed-ring is then able to switch back to the original open-ring form upon visible light irradiation. Also, the two isomers tend not to interconvert in absence of light. This reactive mechanism triggered by the irradiation wavelength which reversibly generate the open or closed-ring isomers is notoriously known as photoswitching. In this way is easy to figure out how different kind of informations can be stored into the two isomers, establishing for example a one-to-one correspondence between the open-ring (closed-ring) geometry and the ON (OFF) state of a device (as shown in Figure 2), creating a molecular switch triggered by light. However the main issue of DTE and many other photochromic systems is the chemical yield which usually results to be very low. This clearly represents a problem for practical application.
![Figure 3. The trans-[Ru(py)4NO]2+ photoisomerization mechanism.](https://mappingignorance.org/app/uploads/2016/10/RuPyNO-640x536.png)
All three isomers possess different physical and chemical characteristics. In particular the well defined photochromism together with the high chemical yield make this molecule a very good molecular switch prototype. Is indeed possible to create a one-to-one correspondence between the two colored isomers and the binary digits 0 or 1. Moreover thanks to the specific absorbing wavelengths, this molecule is also a good candidate for molecular lights sensors and RBG color detectors, to be used in common electronic devices as cell phones or home automation equipments. Besides photoisomerization, nitrosyl metal complexes can also undergo photorelease of NO and thus display biological interest. However this mechanism is still not well understood and further theoretical and experimental investigations are needed to get more insight into the factors controlling the competition between NO isomerization and NO release.
1 comment
[…] El mundo de los nanodispositivos necesita interruptores. Algunos son moléculas que se activan por la luz. Francesco Talotta en Ruthenium nitrosyl complexes: a useful kind of molecular photoswitches […]