The microscopic mechanism behind the vibrational relaxation of adsorbates on metal surfaces
The microscopic mechanism behind the vibrational relaxation of adsorbates on metal surfaces
Most of the theories describing dynamical processes at surfaces rely on the validity of the adiabatic Born-Oppenheimer approximation. What does this mean? We have already seen that adsorption commonly is understood as the reversible binding of molecules and atoms from the gaseous or liquid phase on surfaces, mostly of highly porous adsorbent media. In chemisorption a single layer of molecules, atoms or ions is attached to the adsorbent surface by chemical bonds; in physisorption only van der Waals forces are involved.
The technical applications of adsorption cover a wide range, such as separation, synthesis, storage, purification of gases and liquids, and air conditioning. It is increasingly becoming established in chemical processes as an alternative to the conventional separation techniques of rectification, absorption, condensation, drying, extraction, membrane processes, and catalysis, and it profits from various and optimally tailored adsorbents. It is apparent that all these technical applications are considerably based on a better understanding of the surface science implied.
In the case of gas-surface interactions some approximations are made in order to understand the adsorption process from the molecular point of view. As in the case of purely classical thermodynamic processes, where an ideal irreversible one is considered, meaning that changes are small – infinitesimal – and continuous, a similar assumption is used when considering the quantum approach to the processes involved. This is called an adiabatic approximation.
In any adiabatic approximation, the time dependence of the parameters, say the inter-nuclear distance between the atoms, varies slowly. This means that the solution to the Schrödinger equation at one time goes continuously over to the solution at a later time. The adiabatic approximation was first proposed by Max Born and Vladimir Fock.
An adiabatic approximation in which the motion of atomic nuclei is taken to be so much slower than the motion of electrons that, when calculating the motions of electrons, the nuclei can be taken to be in fixed positions was developed my Max Born and Julius Oppenheimer. The use of the so-called Born-Oppenheimer approximation has been extraordinarily successful in the study of gas-surface elementary processes. However, in metal surfaces low energy electronic excitations are generated by thermal molecules, thus breaking the required smoothness of the adiabatic assumption. But not only molecules would be responsible for altering smoothness.
Enter electrons
In the case of metal surfaces, high concentrations of conducting electrons can, in principle, exchange energy with the adsorbate nuclear degrees of freedom. In fact, there is growing experimental evidence that points out the existence of such non-adiabatic effects. Among them, the significant vibrational line widths of molecules adsorbed on metal surfaces, which are reported in infrared absorption or pump-probe spectroscopy experiments, are considered to be clear fingerprints of the electron-mediated vibrational relaxation. Besides, the non-adiabatic coupling underlies a variety of surface reactions and adsorbate motions (e.g., vibrations, rotations, and lateral hopping) induced by means of inelastic electron currents and femtosecond laser pulses.
Apart from non-adiabatic coupling, a key ingredient for depicting the processes mentioned above is the intermode vibrational coupling. For example, hot-electron induced nonequilibrium dynamics of CO on Cu(100), studied by time-resolved vibrational sum-frequency generation, reveals the importance of intermode coupling in the CO desorption process.
The ensuing theoretical efforts aimed to comprehend these experimental observations are still unable to give precise quantitative estimations of the experimental vibrational relaxation rates and, hence, neither can clarify which relevant mechanisms are ruling them. Everything points to an overlooked relaxation mechanism that should incorporate both the non-adiabatic and intermode couplings on the same footing.
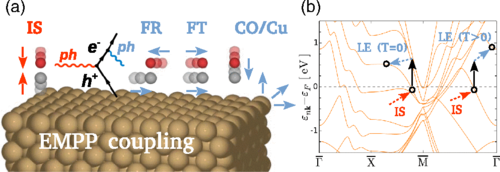
Now, Novko, Alducin and Juaristi demonstrate 1 how non-adiabatic coupling can naturally account for intermode coupling if the former is treated up to second order in the electron-phonon interaction.
The new theory, that includes the process conjoining non-adiabaticity and intermode transitions— electron-mediated phonon-phonon (EMPP) coupling, as the researchers call it— can correctly describe the mechanisms behind the vibrational relaxation of ordered molecules on metal surfaces.
By combining many-body perturbation theory with density functional theory, the researchers investigated a new relaxation mechanism that bridges the non-adiabatic and vibrational intermode couplings, namely, the electron-mediated phonon-phonon coupling, which is then successfully applied to the prototypical case of the CO internal stretch mode on Cu(100). The scientists show that the present phonon line width formula combined with first-principles methodologies is finally able to explain the experimental relaxation rates. Importantly, the results show that the EMPP process dominates over the commonly used first-order non-adiabatic contribution.
Importantly, it is shown that the internal stretch mode couples strongly via electron-hole pairs to other thermally-excited low-energy molecular phonon modes, i.e., to the frustrated rotation and frustrated translation modes. Even more surprising, though, is that surface motion also plays an important role in the electron-mediated vibrational relaxation.
Last, but not least, EMPP explains the temperature dependence of the internal stretch mode line width. This result proves that the temperature dependence is not only triggered by the usual direct phonon-phonon coupling and that the commonly overlooked electron-mediated processes are also a significant factor in this respect.
EMPP is quite general and could be used to elucidate which modes are involved in those surface reactions in which non-adiabatic effects and intermode coupling are expected to be relevant.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance.
References
- D. Novko, M. Alducin, and J. I. Juaristi (2018) Electron-Mediated Phonon-Phonon Coupling Drives the Vibrational Relaxation of CO on Cu(100) Phys. Rev. Lett. doi: 10.1103/PhysRevLett.120.156804 ↩
1 comment
Many proceses at the same time.