Real-time imaging of the forces that build chemical gardens
Real-time imaging of the forces that build chemical gardens
The classic chemical garden experiment is a staple of introductory chemistry, where colorful, plant-like structures sprout from metal salt crystals dropped into a solution of sodium silicate. While these vibrant tubes look like biological life, they are entirely inorganic, driven by the physics of osmosis and the chemistry of precipitation. For decades, scientists have admired these structures, yet the exact internal dynamics of how they grow remained somewhat elusive. A recent study 1 provides a breakthrough by finally letting us see the “invisible” forces at work inside these fragile pipes as they form.
The architecture of a chemical garden

The moment a metal salt crystal (such as manganese sulfate, or commonly copper sulfate or cobalt chloride) hits the silicate solution, the salt begins to dissolve, creating a concentrated pocket of metal ions. This triggers a precipitation reaction at the interface, forming a thin, semi-permeable membrane of metal hydroxide and silica. Because the concentration of dissolved particles is much higher inside this “balloon” than outside, water is drawn in through osmosis. This internal pressure eventually causes the membrane to rupture, usually at the top, where the buoyant force is strongest. As the internal fluid squirts out into the silicate-rich exterior, it reacts again, forming a new section of the tube and repeating the cycle.
Seeing the Invisible: pH and flow
The primary challenge in studying these gardens is their scale and fragility. The membranes are incredibly thin, and the chemistry happens fast. The researchers addressed this by using advanced fluorescence imaging to map the pH and the fluid velocity in real time. They discovered that the interior of the growing tube is highly acidic, typically ranging from below 3.9 up to around 5.9, while the surrounding silicate solution is highly alkaline (commonly pH 11–13). This sharp pH gradient across a membrane roughly one-tenth the width of a human hair is the engine that drives the growth.
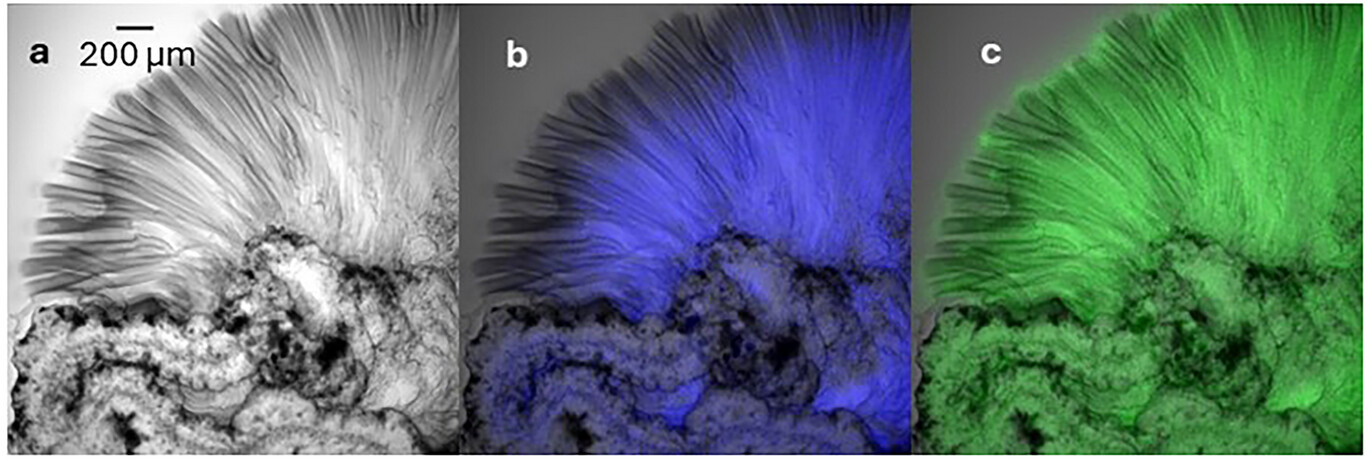
By using pH-sensitive dyes that glow different colors based on the local acidity, the team was able to visualize how these gradients shift during growth. They observed that the acidity is most intense (lowest pH) at the base of the tube where fresh metal ions are released from the dissolving salt, with pH rising toward the growing tip (often reaching above 5.9). Simultaneously, they used microscopic particles to track the “solution flow,” which is the movement of the liquid itself. They found that the flow is not just a simple upward push. Instead, it is a complex interplay between osmotic pressure (which draws water inward, building pressure that pushes the internal fluid out) and buoyancy (which pulls the lighter, acidic fluid upward), including circulation patterns that pump acidic solution toward active growth sites.
Abiotic gradients and life
The ability to image these gradients in situ (in their original place) is more than just a technical feat. It bridges the gap between simple visual observation and quantitative physical chemistry. The study reveals that the membrane acts as a sophisticated chemical reactor. The sharp pH gradient at the membrane wall dictates how quickly the silica precipitates and how “leaky” the membrane becomes. Growth continues as long as sufficient acidity is maintained to keep the membrane permeable and metal ions soluble; when the pH rises too high (around 5.8–5.9) due to inward diffusion of hydroxide ions, permeability decreases, precipitation intensifies, and growth stops or branches deactivate.
Many scientists believe that similar mineral structures at deep-sea hydrothermal vents may have provided the necessary conditions for the origin of life on Earth. These vents also feature steep pH and temperature gradients across mineral membranes, potentially acting as natural reactors or batteries that could have powered the first primitive metabolic reactions. By understanding the real-time dynamics of silica gardens, including how gradients control self-assembly and catalysis, this work offers a clearer window into how such inorganic structures can organize themselves in complex environments and support prebiotic chemistry.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may have been copied verbatim or almost verbatim from the referenced research paper/s.
References
- A. Menichetti, J. Manzi, D. Mordini, F. Otalora, J. M. Garcia-Ruiz, and M. Montalti In situ, real-time imaging of pH gradients and solution flows through silica garden membranes during tube growth Small doi: 10.1002/smll.202507110 ↩