In vivo biochemistry and the definition of pH: a question of scale?
In vivo biochemistry and the definition of pH: a question of scale?
One of the things we learn first, when we get in touch with chemistry, is how important the acidity or alkalinity of a given solution is. This is also and quite often our firstaffair with the chemical equilibrium, because the pH is nothing more than a viewpoint of the water ionization. We were taught that the dissociation of a water molecule is an essential event in acid-base chemistry and the pH definition is based on that process.
It is known that even pure water is subject to auto-ionization because it shows conductivity. More recently, the water auto-ionization has been directly probed by sampling and analyzing ab initio molecular dynamics trajectories 1 . In water auto-ionization, as in every chemical reaction, the equilibrium is reached when the concentrations of reactants and products have no further tendency to change:
2 H2O (l) ⇄ H3O+ (aq) + OH− (aq)
having a water dissociation constant (Kw) defined as:
Kw = [H+][OH-]
where [H+] represents the concentration of hydronium ion (H3O+) and [OH-] the concentration of hydroxide ion (OH-). If both ions have equal concentrations, the solution is neutral. Any impurity or physical disturbance will contribute to shifting this balance, which for pure water at standard conditions for temperature and pressure (25°, 1 atm) is summarized with a Kw=1,0×10−14 and [H+]=1,0×10−7. In an attempt to simplify the acidity of aqueous solutions, Søren Peter Lauritz Sørensen defined the concept of pH in the early 20th century as the negative decimal logarithm of the concentration of the hydronium ion:
pH= -log[H+] = log 1/ [H+]
That results in the pH scale, ranging from 1 to 14, where a neutral solution has pH = 7, an acidic one has pH <7, and a basic one has pH> 7.
The above definition of pH has been valid and accurate for ages in those sciences working with solutions on a daily basis, but it could fail when it faces chemistry at life´s level. Since the pH definition depends on the concept of Kw constant and the volume of the compartments at cellular or subcellular scale range from picoLiters (pL, 10-15 m3) to attoLiter (aL, 10-21 m3), a Gedankenexperiment can be done as follows.
The Kw constant under standard conditions is a pretty small number, but it is not much bigger at 37°C, which means that only very few water molecules will show dissociated state under those conditions. Using this constant, the minimal volume of water required to find at least one of the water molecules dissociated in ions (H+ andOH-) under a given condition of pH and temperature can be calculated. In other words, the following formula is able to relate pH to the minimal volume required to find ions derived from water dissociation.
VpH=10(pH-pKw/2) x (10pKw/2 /NA)
here NA is Avogadro’s number and VpH the minimal volume required.
Therefore, using the above formula, the VpH for a given pH is calculated. Then, those VpH values are compared with the volume and the reported biological pH for several kinds of cells or subcellular organella (Fig.1).

What emerges from the comparison is a conflict with two sides, which the authors of the work in question 2 have called the ‘‘hard’’ (Fig.1, grey zone) and the ‘‘soft pH crisis”(Fig.1, white zone). The “hard crisis” rises when the volume of a biological structure is inconsistent with its reported pH values considering the VpH values. E.g. for lysosomes the average size is around 30aL and the reported pH measured by fluorescent sensors is about 4´8. According with the current Kw constant definition, the minimum volume to get one single water molecule dissociated at pH 4´8 is about 10fL, which is more than a thousand times bigger than the reported one. This means that following current Kw understanding, such subcellular compartments should oscillate between ‘‘pH’’ and ‘‘no pH’’ states, which is highly improbable due to the importance of H+ ions in biological reactions. The “soft crisis” is not much softer, because, in the light of the calculations, it seems that even cells or compartments like mitochondria would dispose only a few hundreds or thousands of protons in order to perform countless acid-base reactions and generate gradients to push ATPases and other pump-like molecular machines.
Two arguments are introduced to explain such discrepancies between the pH reported by chemical sensors and the pH in biological attoliter volumes according to the Kw, and therefore in accordance with the pH definition. One of those arguments affect directly to the water equilibrium and Kw constant validity in extremely small volumes. Pointing that hydronium ions (H3O+) and hydroxide ions (OH-) would be no longer in equilibrium with H2O. Meaning this that the Kw is no more valid as a thermodynamic constant at nanometric scale.
On the other side, the chemical sensors in question, roughly defined, just to be molecules with protonable groups which change their spectrophotometric properties once they are located in an environment with a pH close to their own pKa (e.g. Fig.2).
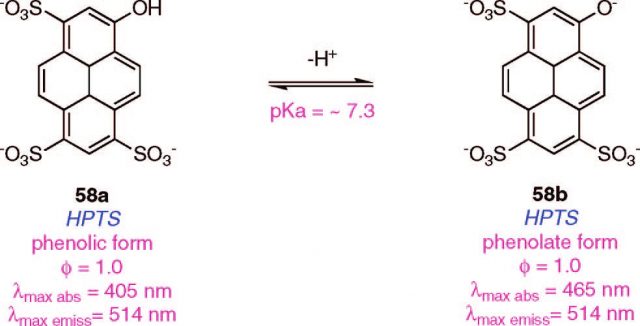
The probe protonation is supposed to happen by H+ protons proceeding from the water equilibrium (as it does in milliliter scale calibration experiments were the probe is in significant excess in relation with the analyzed protons). Therefore, a characteristic change on its absorption or emission spectra can be measured if the compound reach a compartment with an adequate pH. Letting aside that those probes will hardly be in excess when filling a femtoliter volume, the second explanation goes on to assume a change in the definition of what molecular probes interpreted as pH.
Another water independent source of protons must exist to allow not only the expectral change of those molecular sensors, but all the other cellular acid-base reactions and proton gradients. It is proposed that the in vivo acid-base chemistry could occur by pairwise direct proton and hydroxide exchange between biomolecules. Some good examples of biomolecules which could fulfill that gap are inorganic phosphate, nucleotides and membrane phospholipids. All of them with pK values around 7. Summarizing, what these chemical pH sensors would measure is the equilibrium constant of a direct bimolecular (donor/ acceptor) reaction.
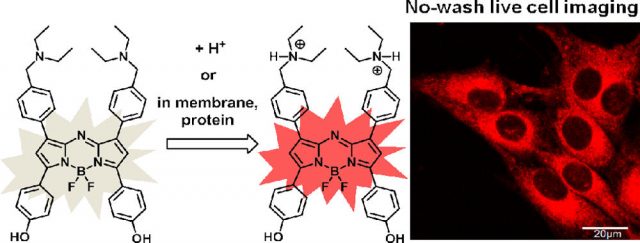
In this scenario, rising from a gedankenexperiment, the later explanation seems to be more probable. It contributes to explain why chemically different sensors report different pH for the same compartment as well as some observed experimental variability. Furthermore, a recent research about the intracellular behavior of some pH probes called aza-BODIPY dyes 3 provides experimental evidence supporting indirectly the second hypothesis. After an exhaustive research it is concluded that the change in their spectrophotometric properties is not due to the compartment pH, but because of their pKa shift when they become incorporated in membranes and proteinic environment (Fig. 3). Note that, the pKa shift of a chemical sensor located in lipidic or proteinic environment do not probe the experimental validity of the above calculations. However, it could point out that, in fact, this in vivo pH sensors do not feel the pH (the H+ concentration) but their availability in donor/acceptor reactions, which parameters (e.g. pKa) are highly dependent on the local conditions. More experimental work has to be dome to resolve the disagreement, because it could imply a change not only in the pH definition but in the current conception of the actual in vivo biochemistry.
References
- Geissler, P. L., Dellago, C., Chandler, D., Hutter, J. & Parrinello, M. Autoionization in Liquid Water. Science 291, 2121–2124 (2001) ↩
- Bal, W., Kurowska, E. & Maret, W. The Final Frontier of pH and the Undiscovered Country Beyond. PLoS One7, (2012) ↩
- Zhang, X.-X. et al. pH-Sensitive Fluorescent Dyes: Are They Really pH-Sensitive in Cells? Mol. Pharmaceutics (2013). doi:10.1021/mp3006903 ↩
3 comments
Wow, a very interesting but complicated story. Once again, what we think we know about the cell turns out to be more and more complex… who knows what kind of knowledge we will have some decades from now.
If you pardon me, I’m going to remove the figure of a manuscript I’m working with in which we talk about optimum pHs and stuff like that… O_o
Well, according to those recent findings, It would not be quite correct to talk about intracellular optimum pHs but the optimum condition which permit an equilibrium constant of a direct donor/ acceptor reaction compatible with life. Slightly different but with an enormous change in the biochemistry conception.
The Final Frontier of pH and the Undiscovered Country Beyond.
The calculations in this article are wrong.
First I got a soft crisis, but after calculating it again I got a hard crisis.