Cellulose conversion to starch, a promising strategy for future global food demand
Cellulose conversion to starch, a promising strategy for future global food demand

Carbohydrates are molecules composed of carbon, oxygen and hydrogen. They are formed of at least three atoms of carbon. Monosaccharides are the simplest carbohydrates and they cannot be hydrolyzed to smaller carbohydrates. Among the monosccharides we find glucose or fructoses which are formed of six atoms of carbon or glyceraldehyde which is composed of three atoms of carbon atoms. The binding of monosaccharides leads to the formation of more complex molecules. Among them, dissacharides are the simplest ones; they are formed out of the glycosidic linkage of two monosaccharides. For example, sucrose is a dissacharide created from the binding of glucose and fructose. On the other hand, polysaccharides are very long chains of monosaccharides composed from hundreds to thousands of sugars like cellulose, starch, quitin or glycogen.
Carbohydrates are part of animals main dietary components. About the 50% of the daily energy that a person needs comes from carbohydrates and their main function is to provide energy for the body. Nevertheless, it is worth saying that they are not essential since the body can get the energy he needs from proteins and lipids. The United Nations Food and Agriculture Organization (FAO) advices that up to three quarters of our diet should be based of carbohydrates. Among these, starch is the most important one. Starch is a storage polyssacharide and it accounts for more than half of the consumed carbohydrates, providing up to 60% of the energy needed by humans. Starch is a long chain of glucose molecules which are bind by alpha-1,4-glycosidic bonds and alpha-1,6-glycosidic bonds. Starch is produced by photosynthetic plants as energy store and some plants contain it in large quantities like potatoes, wheat, rice or maize. Starch can be of two types, amylose which is a linear molecule or amylopectin which is branched.
Cellulose is a structural polyssacharide, it is the supporting material of plant cell wall and it represents the most abundant carbohydrate on Earth. It is formed of a linear glucan linked by beta-1,4-glycosidic bonds. Animals, cannot directly use cellulose as source of energy since they lack cellulase, an essential enzyme for cellulose breakdown. Some animals like rummiants (cow, sheep, etc.) and insects like termites have established stable mutualistic relationships with microorganisms that live inside their digestive systems that are able to hydrolyze cellulose and make the glucose available for them. Annual resource of cellulosic materials is ∼40 times greater than the starch produced by crops cultivated for food and feed. In addition, (perennial) cellulosic plants require fewer inputs such as fertilizers, herbicides, pesticides, and water, whereas annual high-productivity starch-rich crops require high-quality arable land, enough water, and high inputs. Besides, every ton of cereals harvested is usually accompanied by the production of two to three tons of cellulose (You et al., 2013).
The research group of Y.H. Percival Zhang of the Department of Biological Systems Engineering of the Virginia Polytechnic Institute and State University recently published in the Procedings of the National Academy of Sciences of the United States of America an efficient in vitro enzymatic conversion of cellulose to starch which, in their opinion, could play an important role in the actual context of the exponential global food demand 1. To do so, the authors designed a cell-free biosystem composed of a synthetic enzymatic pathway that can transform solid cellulose into amylose. This process is called simultenous enzymatic biotransformation and microbial fermentation (SEBF) and it is formed of two steps: cellulose hydrolysis and amylase synthesis (Fig. 1).
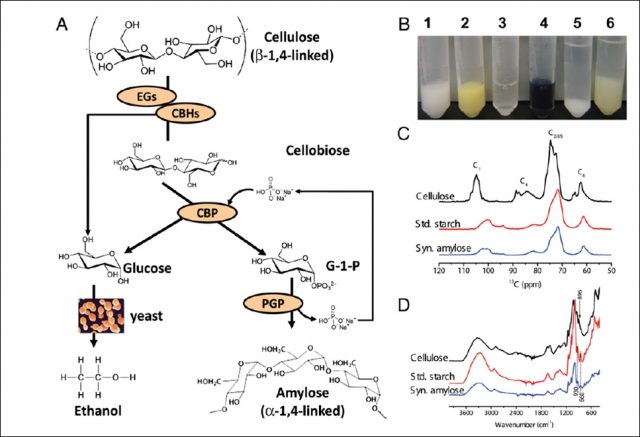
For the first step, You and collaborators tested five different cellulases from different organisms and found the best cellulose hydrolysis yield with the use of a Bacillus subtilis endoglucanase (EG) and a cellobiohydrolase (CBH) from the fungus Trichoderma. For the second step, the synthesis of amylose, the authors purified a cellobiose phosphorylase (CBP) from the bacterium Clostridium thermocellum and after testing several alpha-glucan phosphorylases from different organisms they chose the one coming from potato plants (PGP) for being the most suitable one for amylose synthesis in vitro. With this SEBF method, the obtained a yield of 14,4% (wt/wt), this means 0.144g of amylose per gram of cellulose. After adding glucose oxidase to remove glucose, which is known to inhibit CBP activity, the yield increased up to 30%. Moreover, in this system developed by You et al. to synthesize starch, non-used glucose molecules generated from cellulose hydrolysis and CBPs can be assimilated by ethanol-producing yeast Saccharomyces cerevisiae. This process is easy to scale up for commercial production and it does not require expensive equipment or chemical reagents. Moreover the authors improved a system for the enzymes recycling by the used of magnetic nanoparticles.
Synthetic amylose has a great variety of applications. Among others, it can be used to make biodegradable plastics (starch-based plastics represent 50% of the bioplastics market). Top-quality amylose can be used, for example, for drug capsules in the pharmaceutical industry. High-quality amylose can serve to manufacture transparent and flexible plastic sheets and films. Amylose is also used in the food industry as emulsion stabilizer and gelling agent. Food-grade amylose can be used as tailored food for meeting special dietary requirements. Besides, medium-quality amylose can serve as a high-density hydrogen storage carrier that could solve problems related to hydrogen storage and distribution and low-quality amylose can be used for animal feed. Thus this system represents a promising strategy for the use of nonfood agricultural and forest residues.
References
- You C, Chen H, Myung S, Sathitsuksanoh N, Ma H, Zhang X-Z, Li J and Zhang YHP (2013) enzymatic transformation of nonfood biomass to starch. Procedings of the National Academy of Sciences of the United States of America doi: 10.1073/pnas.1302420110. ↩