Lipids and autism
Lipids and autism
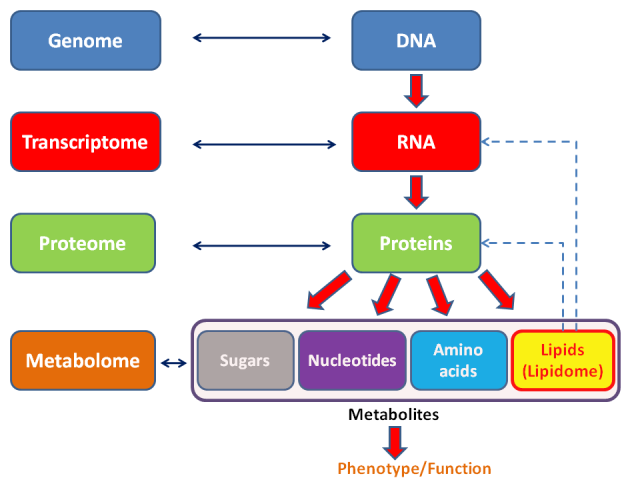
Lipids represent one of the fundamental molecular components within the realm of biological organisms, alongside proteins, carbohydrates, and nucleic acids. The concept of the “lipidome” pertains to the entirety of lipids found within a cell or a collection of cells. This terminology emerged within the context of modern biological sciences, often referred to as “omic sciences,” which encompasses the genome (DNA), transcriptome (mRNA), microbiome (microorganisms inhabiting the human body), and proteome (proteins), among others. Analyzing the lipidome involves techniques such as mass spectrometry, bioinformatics, and conventional laboratory methods.
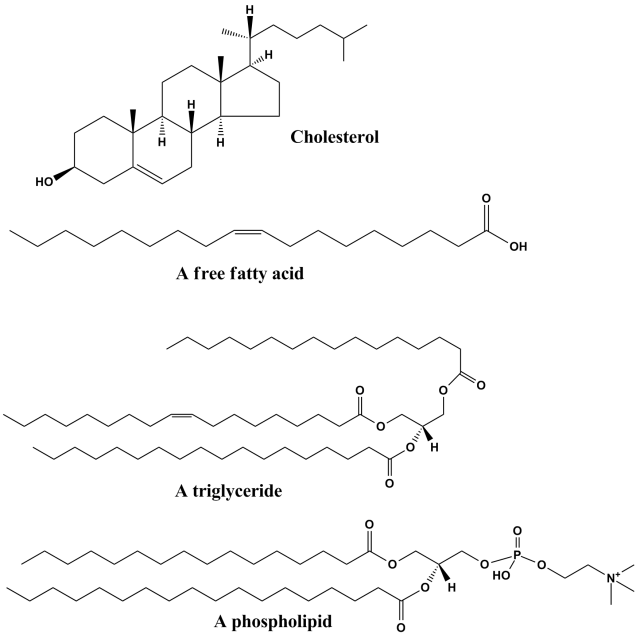
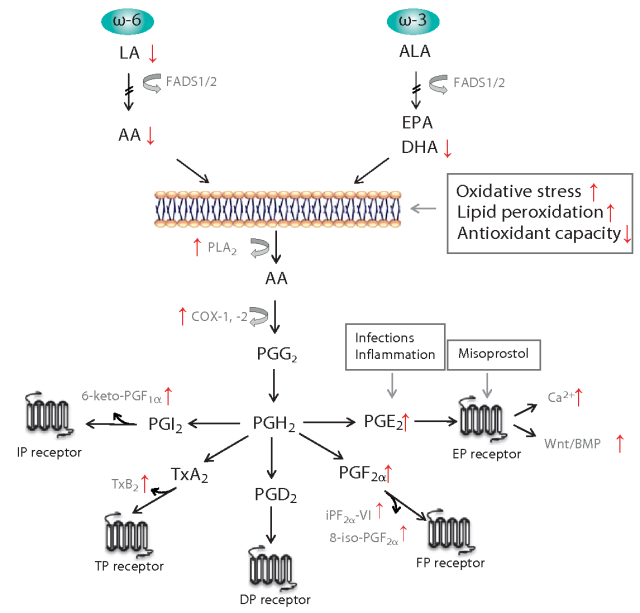
Lipids serve as pivotal regulatory molecules, actively contributing to the growth and maintenance of brain functions. Perturbations in fatty acid metabolic pathways could potentially play a role in the development of Autism Spectrum Disorder (ASD). One natural lipid-derived compound, prostaglandin E2, is implicated in the physiological processes associated with ASD. Specifically, esterified omega-6 polyunsaturated fatty acids (PUFAs), such as arachidonic acid (AA), located on the inner surface of cell membranes, undergo conversion by phospholipase A2 into their free form. Subsequently, these free fatty acids are further metabolized by enzymes like cyclooxygenases and lipoxygenases to yield bioactive mediators, including prostanoids and leukotrienes. These mediators have emerged as potential novel targets for both preventive and therapeutic approaches in addressing inflammatory conditions, including specific forms of ASD 1.
.
In human plasma, the lipidome comprises nearly 600 distinct molecular species, and its unique characteristics can provide insights into the potential risks associated with certain diseases, like cancers linked to dietary fats, notably breast cancer. In the field of autism, rare genetic variations in lipid metabolism genes (e.g., EFR3A4) are associated with non-syndromic autism spectrum disorders (ASD), and hereditary lipid metabolism syndromes often coincide with neurodevelopmental delays (e.g., Smith-Lemli-Opitz syndrome, Niemann-Pick syndrome, and Tay-Sachs syndrome). Beyond these infrequent genetic diagnoses, researchers have explored the broader connections between lipids and ASD, yielding mixed findings. Some smaller-scale studies have suggested potential links between low cholesterol levels and ASD diagnoses, while a comprehensive investigation integrating genetic and health record data has indicated the existence of an ASD subtype marked by abnormal lipid profiles. Notably, individuals with autism appear to be at an elevated risk for treatable cardiometabolic diseases.
A study recently published in Nature Medicine 2 has uncovered a potential causal connection between aspects of the lipidome (specifically reduced levels of long-chain polyunsaturated fatty acids in blood plasma) and sleep quality issues and sleep disorders observed in autistic children. The research involved identifying lipids associated with ASD diagnosis (n = 8), sleep disturbances (n = 20), and cognitive function (n = 8). Additionally, a myriad of lipid species-level associations were observed in conjunction with age (n = 181), Tanner stage (a scale measuring physical development) (n = 43), body mass index (n = 159), and sex (n = 71). Notably, the study also found that the lipidome characteristics in autistic children experiencing sleep disturbances bore similarities to disturbances often linked to an unhealthy diet. This research was conducted using the Australian Autism Biobank, ensuring a substantial sample size of autistic individuals and controls.
One plausible explanation for a portion of these findings relates to the selective dietary habits commonly observed in individuals with autism. The researchers discovered significant interactions between the ASD lipidome profile, ASD diagnosis, and reduced meat consumption, indicating that the lipidome profile in individuals with ASD may be influenced, in part, by lower meat consumption.
The researchers meticulously analyzed a diverse panel of 800 different fats present in the blood plasma of 765 children, with 485 of them having been diagnosed with autism. They identified a potential causal link between decreased levels of long-chain polyunsaturated fatty acids (some of which are commonly found in fish oil, eggs, and meat) and poor sleep quality. The lipidome characteristics of children experiencing sleep problems closely resembled the patterns associated with an unhealthy diet, shedding light on a potential mechanism through which sleep issues contribute to adverse health outcomes. However, it is important to note that further research is necessary before making specific dietary recommendations.
Dr. Chloe Yap, the lead author of the study, emphasized that this research marks a significant shift in autism research focus. While past biological research in autism primarily centered on enhancing diagnostic methods, this study delves into often overlooked areas, like co-occurring conditions that significantly impact the quality of life of autistic individuals, such as sleep disturbances and feeding issues.
In conclusion, the field of lipidomics provides valuable insights into the intricate nature of neurodevelopment and the biological ramifications of conditions that frequently affect the well-being of autistic individuals. The next step in this research is to explore the connections between sleep, diet, and traits associated with autism during childhood. This will enhance our understanding of the factors influencing brain development and, consequently, their impact on various life domains, including education, employment, and overall quality of life.
References
- Yui K, Imataka G, Yoshihara S (2022) Lipid-Based Molecules on Signaling Pathways in Autism Spectrum Disorder. Int J Molec Sci 23(17): 9803. ↩
- Yap CX, Henders AK, Alvares GA, Giles C, Huynh K, Nguyen A, Wallace L, McLaren T, Yang Y, Hernandez LM, Gandal MJ, Hansell NK, Cleary D, Grove R, Hafekost C, Harun A, Holdsworth H, Jellett R, Khan F, Lawson LP, Leslie J, Levis Frenk M, Masi A, Mathew NE, Muniandy M, Nothard M, Miller JL, Nunn L, Strike LT, Cadby G, Moses EK; Busselton Health Study Investigators; de Zubicaray GI, Thompson PM, McMahon KL, Wright MJ, Visscher PM, Dawson PA, Dissanayake C, Eapen V, Heussler HS, Whitehouse AJO, Meikle PJ, Wray NR, Gratten J (2023) Interactions between the lipidome and genetic and environmental factors in autism. Nat Med 29(4): 936-949. doi: 10.1038/s41591-023-02271-1 ↩
1 comment
[…] post Lipids and autism appeared first on Mapping […]