Engineering nitrogen-fixing cereals, between science fiction and reality
Engineering nitrogen-fixing cereals, between science fiction and reality
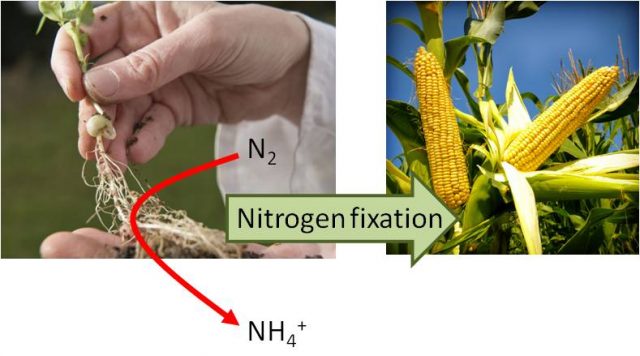
Every organism, from virus to animals need nitrogen (N) to live; indeed, nucleic acids and proteins are made out with N. Nearly all of these organisms, fulfill their nitrogen needs taken up N from the environment in reduced forms, usually as nitrate (NO3–), ammonium (NH4+) or amino acids. Interestingly, certain microorganisms, known as diazatrophs, have developed through evolution the ability to use atmospheric nitrogen (N2), which represents around 78% of the atmosphere, for their growth and development. This reaction is exclusively catalyzed by a complex and extremely oxygen-sensitive enzyme called nitrogenase. Interestingly, some plants, mainly those belonging to the family of legumes have acquired the capacity to develop a symbiotic relationship with bacteria that derives on the apparition of a new organ, the nodule, where bacteria fix nitrogen (convert N2 to NH4+), and export it to the plant. In exchange, the plant provides bacteria with a “house”, nodules, with microaerobic conditions to allow nitrogenase functioning and with energy and carbon obtained by the plant photosynthesis.
This capacity of legumes has been used in agriculture as a way to introduce N into the system by the traditional practice of crop rotation, commonly between cereals and legumes. However, in intensive agriculture, the need to maximize crops yield makes necessary the supplementation of soils with additional N, usually as chemically synthesized fertilizers. These fertilizers are very costly and for farmers of undeveloped countries even impossible to buy. Besides, N fertilization is an important source of contamination. Indeed, the industrial Haber-Bosch nitrogen fixing process, first used in 1913, requires the consumption of huge quantities of fossil fuels, which represents around 2% of the world´s energy consumption. Moreover, once in the soil an important part of the N supplied is lost, part of it due to nitrate leaching, which provokes water contamination, and part of it is also lost to the atmosphere among others as nitrogenous gases, notably in form of N2O, which is an important greenhouse gas. Therefore, maintaining crops yield with the minimum N supply is an important challenge for future agriculture. In this context, since legumes don´t need external N supply to grow, the idea of trying to transfer their nitrogen fixing capacity to important non-fixing crops, such as wheat or maize, is one of the long-standing dreams of many researchers in plant biology.
Although transferring nitrogen fixation to cereals is still considered one of the hugest challenges in plant biology, the recent development of genetic engineering tools allowing the construction of large multigene synthetic cassettes and the emergence of efficient and affordable DNA synthesis has made easier the transfer o genes. Moreover, the important support of “Bill and Melinda Gates foundation” that started to fund projects in this research area coordinated by Prof. Gyles Oldroyd from the John Innes Centre in Norwich (UK), has made researchers are shedding some new light on this topic. Indeed, although some decades will be needed, the research community is starting to think that it will be reasonably possible to achieve the challenge.
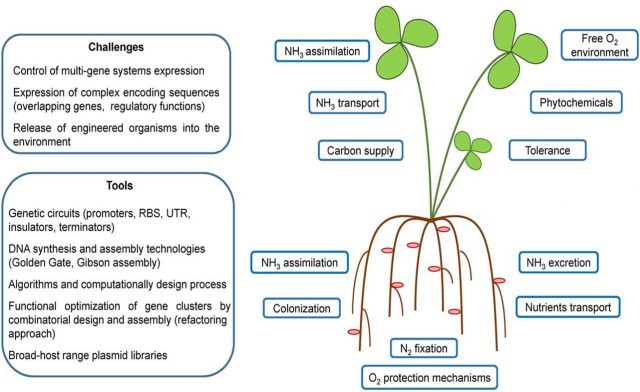
The research engaged is focused in three different strategies 123. A first approach is to transfer the genes of legume plants necessary for the development of root nodule symbiosis to cereals. A second approach is to create nodule-independent nitrogen-fixing cereals promoting their association with endophytes that fix nitrogen. The third approach is to directly introduce nitrogenase into the plant.
Regarding the first approach, to transfer legume symbiotic nitrogen fixation to cereals three steps need to be engineered: bacteria perception, bacterial infection and appropriate nodule organogenesis compatible with nitrogenase activity. In this sense, important advance are being made in the understanding of the legume-rhizobial symbiosis and in cereal symbiosis with arbuscular mycorrhizal fungi. Indeed, both processes share common signaling elements that will be essential to make easier this difficult approach.
Concerning the second approach, it involves the manipulation of both partners and is mainly based in transferring nitrogen fixing capacity to bacteria that already associate with cereals. In this sense nitrogen fixation cassettes built using synthetic biology has been successful transferring nitrogen fixation to the facultative anaerobe E. coli 4. However, to be effective this should be achieved in natural colonizers/endophytes of cereals that are aerobic and of course the ammonia should be excreted outside the bacteria to be used by the plant 5.
Finally, the third approach is probably the one that is giving more promising results. Indeed, one of the research groups working in directly transferring nitrogenase to plants, led by Prof. Luis M. Rubio in the Polythecnic Univeristy of Madrid recently published the expression active nitrogenase Fe protein in the mitochondrial matrix of yeast 6 and the formation of nitrogenase tetramers by transferring 9 nif genes also to yeast mitochondria 7; thus, showing the convenience of this organelle to overtake the limitation of nitrogenase oxygen sensitivity.
In conclusion, engineering artificial symbiosis or associations leading to the development of nonlegume nitrogen-fixing crops would be a huge advance to reduce the global demand of nitrogen fertilizers. Although still far from the goal, the major discoveries and technical advances achieved in recent years encourage the pursuit of this titanic challenge.
References
- Beatty PH, Good AG (2011) Plant Science. Future prospects for cereals that fix nitrogen. Science. doi: 10.1126/science.1209467. ↩
- Rogers C, Oldroyd GED (2014) Synthetic biology approaches to engineering the nitrogen symbiosis in cereals. Journal of Experimental Botany. doi:10.1093/jxb/eru098 ↩
- Mus F, Crook MB, Garcia K, Costas AG, Geddes BA, Kouri ED, Paramsivan P, Ryu M-H, Oldroyd GED, Poole PS; Udvardi MK, Voigt CA, Ané J-M, Peters JW (2016) Symbiotic nitrogen fixation and the challenges to its extension to nonlegumes. Applied and Environmental Microbiology. doi:10.1128/AEM.01055-16. ↩
- Temme K, Zhao D, Voigt CA (2012) Refactoring the nitrogen fixation gene cluster from Klebsiella oxytoca. Proceeding of the National Academy of Sciences. USA doi:10.1073/pnas.1120788109 ↩
- Geddes A, M-H, Mus F, Costas AG, Peters JW, Voigt CA and Philip Poole P (2015) Use of plant colonizing bacteria as chassis for transfer of N2-fixation to cereals. Current Opinion in Biotechnology. doi:10.1016/j.copbio.2015.01.004 ↩
- López-Torrejón G, Jiménez-Vicente E, Buesa JM, Hernandez JA, Verma HK, Rubio LM (2016) Expression of a functional oxygen-labile nitrogenase component in the mitochondrial matrix of aerobically grown yeast. Nature Communications. doi:10.1038/ncomms11426. ↩
- Burén S, Young EM, Sweeny EA, Lopez-Torrejón G, Veldhuizen M, Voigt CA, Rubio LM (2017) Formation of Nitrogenase NifDK tetramers in the mitochondria of Saccharomyces cerevisiae. ACS Synthetic Biology. doi:10.1021/acssynbio.6b00371 ↩
2 comments
[…] hori nitrogenoa finkatzeko gai diren zerealak diseinatzeko? Daniel Marinok azaldu du asteon Engineering nitrogen-fixing cereals, between science fiction and reality […]
[…] Si las legumbres son capaces de fijar el nitrógeno, ¿por qué no usar esa capacidad para diseñar cereales que sean capaces de hacerlo? Sería una auténtica revolución económico-agrícola-alimentaria. Daniel Marino nos lo explica en Engineering nitrogen-fixing cereals, between science […]