Why would pressure stretch a chemical bond?
Why would pressure stretch a chemical bond?
Pressure is usually imagined as a force that simply squeezes matter into a smaller space. At the molecular level, however, the story is more subtle. A recent computational chemistry study 1 explored how pressure changes the shapes of molecules and the fleeting structures known as transition states, which appear briefly as reactions occur. The work focused on pressures up to about 5 gigapascals (roughly fifty thousand times higher than atmospheric pressure) but still within the range used in high-pressure chemistry experiments.
The central question was simple: when molecules are compressed, which parts of them change the most? The answer turned out to depend on three main factors: how stiff a bond is, how much the molecular volume changes when atoms move, and whether the structure is a stable molecule or a transition state.
Microscopic effects of high pressure
To investigate this, the researchers used a computational method called the Extreme-Pressure Polarizable Continuum Model, or XP-PCM. Instead of treating pressure as a direct mechanical push on atoms, the model surrounds a molecule with a compressed environment that mimics matter under pressure. As that space shrinks, the molecule’s own electrons are repelled more and more strongly by those of the surrounding medium, faithfully reproducing the microscopic effects of high pressure.
The calculations showed that strong covalent bonds resist pressure remarkably well. In methane and benzene, ordinary carbon–hydrogen and carbon–carbon bonds shortened by less than 0.01 ångström between 0 and 5 GPa. An ångström is one ten-billionth of a metre, so these changes are extremely small. Strong bonds behave like stiff springs: pressure can compress them only slightly.
Weaker interactions responded much more dramatically. In diborane, which contains unusual electron-deficient bonds, the weaker bridging bonds compressed more than ordinary boron–hydrogen bonds. Even larger changes appeared in systems held together mainly by nonbonded interactions, where there is no strong chemical bond resisting compression. In molecules such as cyclophane, where two benzene rings sit close together without forming direct covalent bonds, the inter-ring distance shrank by about 0.05 to 0.07 ångström over the same pressure range. Weak interactions are therefore far more compressible than strong covalent bonds.
Metal-containing molecules displayed intermediate behaviour. In ferrocene, an iron atom sits between two cyclopentadienyl rings like a sandwich filling. Under pressure, the iron–carbon distances shortened noticeably more than ordinary covalent bonds but less than weak nonbonded contacts. The calculations also reproduced an experimentally observed pressure-induced structural change in ferrocene, showing that the computational approach captures real molecular behaviour accurately.
One of the most interesting aspects of the study involved transition states. Transition states are unstable arrangements of atoms located at the top of the energy barrier separating reactants and products. Because reactions pass through them only briefly, they are difficult to study experimentally. Pressure can change their geometries in surprising ways.
Why would compression stretch a bond?
In most stable molecules, pressure shortens bonds. Yet some transition states behaved in the opposite manner. In the transition state of the Diels–Alder reaction between butadiene and ethene, the forming carbon–carbon bonds actually became longer under pressure. At first this seems counterintuitive: why would compression stretch a bond?
The explanation comes from the shape of the reaction’s energy landscape. Pressure lowers the reaction barrier, shifting the transition state toward an earlier stage of the reaction. In that earlier structure, the reacting molecules are farther apart, so the forming bonds are longer. Rather than simply squeezing every bond equally, pressure changes where the transition state sits along the reaction pathway.
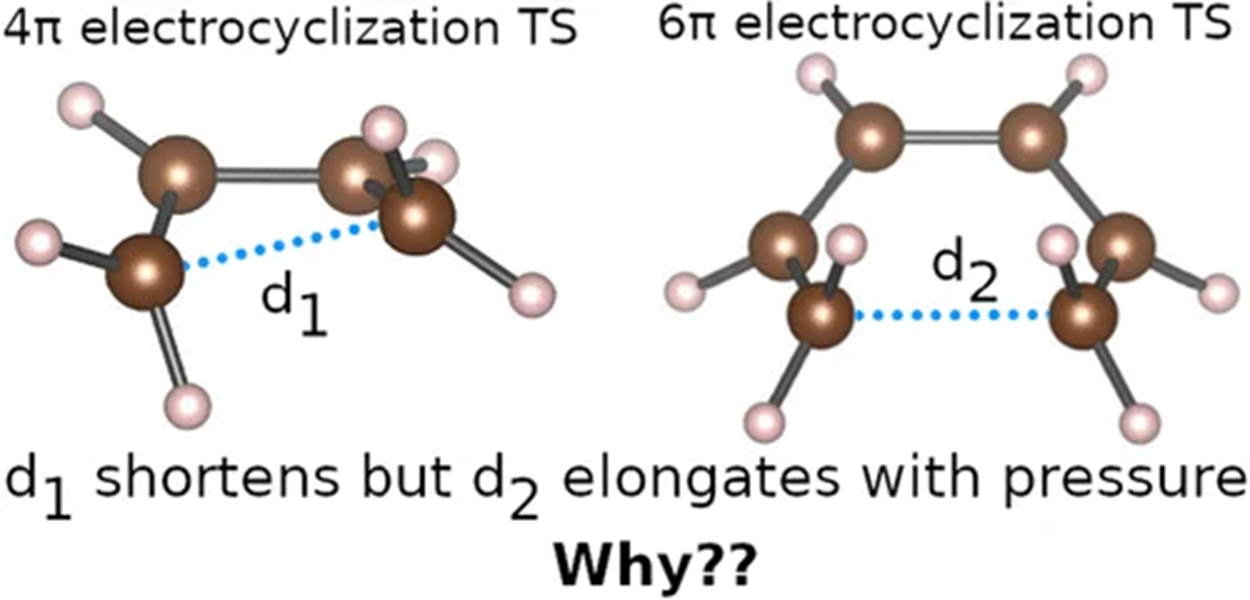
Other reactions showed different behaviour. In some electrocyclic reactions, pressure lengthened key transition-state distances, while in others it shortened them. The outcome depended on the precise atomic motions involved in the reaction. If the important motion mainly involved bond stretching, pressure tended to favour elongation in the transition state. If rotational motions dominated instead, ordinary bond compression could outweigh the stretching effect.
The study also examined hydrogen-transfer reactions and the Cope rearrangement, a classic organic reaction involving simultaneous bond breaking and bond formation. In these cases, pressure significantly reduced certain long, weak transition-state distances. For the Cope rearrangement, the compression was especially large — up to 0.2 ångström at 5 GPa — comparable to the structural change caused by adding certain chemical substituents to the molecule.
A key feature of the work was its mode-by-mode analysis. Molecules vibrate in characteristic patterns called normal modes. By examining how pressure affects each vibrational mode separately, the calculations identified exactly which molecular motions control structural changes under compression. In ferrocene, for instance, the mode that brings the two rings closer to the iron proved far more sensitive to pressure than the stiffer carbon–hydrogen stretching. Soft vibrations associated with weak interactions were most sensitive to pressure overall, while stiff stretching modes of strong covalent bonds changed little.
Overall, the study established a clear hierarchy. Strong covalent bonds are least affected by pressure, metal–ligand bonds are more compressible, and weak nonbonded interactions are the most sensitive of all. Transition states add another level of complexity, because pressure can sometimes push them toward structures with unexpectedly longer bonds.
These results offer more than a catalogue of compressed molecules. They provide a framework for predicting how chemical reactions and molecular structures behave in extreme environments, from deep inside planets to high-pressure industrial chemistry. Pressure is not merely a tool for squeezing matter closer together. It can reshape energy landscapes, redirect reaction pathways, and reveal unexpected features of chemical bonding itself.
Author: César Tomé López is a science writer and the editor of Mapping Ignorance
Disclaimer: Parts of this article may have been copied verbatim or almost verbatim from the referenced research paper/s.
References
- Jorge Laranjeira, Ruobing Lang, Roberto Cammi, and Bo Chen (2026) Effect of Pressure on Molecular and Transition-State Geometries Journal of Chemical Theory and Computation doi: 10.1021/acs.jctc.5c02067 ↩